To describe human papillomavirus (HPV) vaccination coverage among people living with human immunodeficiency virus (HIV) and evaluate factors influencing completion of the vaccination schedule.
MethodRetrospective study of 198 HIV patients aged ≥18 years in active follow-up at Hospital Clínico Universitario San Juan (Alicante, Spain) during 2018–2023. Sociodemographic and clinical variables, referral/attendance at preventive medicine service (PMS), and completion of HPV vaccination (3 doses) were collected. Adjusted logistic regression analyses were used to determine factors associated with complete HPV vaccination.
ResultsOverall, 19.7% (39/198) of individuals in follow–up successfully completed the three–dose HPV vaccination regimen. In the multivariable model adjusted for age, biological sex, CD4 nadir, time since HIV diagnosis, and time in study, attendance at PMS (OR: 4.20; 95%CI: 2.00–8.80), time since diagnosis (OR: 1.19; 95%CI: 1.00–1.42), and loss to follow-up (OR: 0.14; 95%CI: 0.04–0.33) were associated with complete vaccination.
ConclusionsHPV vaccination coverage among people with HIV remains suboptimal. Referral and attendance at PMS are key determinants of vaccination success, whereas loss to follow–up represents a major barrier. Interventions in tertiary care should prioritize patient retention after referral to specialized programs.
Describir la cobertura vacunal frente al virus del papiloma humano (VPH) en personas que viven con infección por el virus de la inmunodeficiencia humana (VIH) y evaluar los factores que influyen en completar el esquema de vacunación.
MétodoEstudio retrospectivo de 198 pacientes VIH positivos ≥18 años en seguimiento activo en el Hospital Clínico Universitario San Juan (Alicante, España) en el periodo 2018-2023. Se recopilaron variables sociodemográficas, clínicas, derivación/asistencia al servicio de medicina preventiva (SMP) y vacunación completa frente al VPH (3 dosis). Se utilizaron regresiones logísticas ajustadas para determinar los factores que se asociaron con la vacunación completa frente al VPH.
ResultadosLa cobertura de vacunación completa fue del 19,7% (n=39/198). En el modelo multivariable ajustado por edad, sexo biológico, CD4 nadir, tiempo desde el diagnóstico de VIH y tiempo en estudio, la asistencia al SMP (OR: 4,20; IC95%: 2,00-8,80), el tiempo desde el diagnóstico (OR: 1,19; IC95%: 1,00-1,42) y ser perdido durante el seguimiento (OR: 0,14; IC95%: 0,04-0,33) se asociaron con vacunación completa.
ConclusionesLa cobertura de vacunación frente al VPH en personas con VIH es subóptima. La derivación y la asistencia al SMP son determinantes clave, pero las pérdidas de seguimiento limitan el éxito. Las intervenciones en atención terciaria deben priorizar la retención de pacientes tras la derivación a programas especializados.
People living with human immunodeficiency virus (PLWH) face an increased risk of persistent human papillomavirus (HPV) infection and progression to HPV-related neoplasms such as cervical, anal, and oropharyngeal cancers. This heightened risk reflects both increased exposure to HPV and impaired immune surveillance of infected cells, particularly in those with advanced immunosuppression.1
Guidelines from the European AIDS Clinical Society (EACS) recommend HPV vaccination for people living with HIV (PLWH) regardless of sex. In Spain, the Grupo de Estudio del SIDA (GESIDA) and the Ministry of Health recommend HPV vaccination for PLWH up to 26 years of age, with vaccination offered selectively between 27 and 45 years based on individual risk assessment.1–3 The Spanish Ministry of Health in 2018 published comprehensive guidelines on vaccination in high-risk populations, identifying HPV vaccination as a priority preventive service for PLWH with an age cutoff at 26 years for routine vaccination and up to 45 years for individualized risk assessment.4 While vaccination is safe and immunogenic in this group, antibody titters are lower than in HIV-negative people, with weaker immune responses in those with advanced immunosuppression or detectable viremia.5,6 Vaccine safety has shown no harmful effects on HIV viral load or CD4 counts, and serological responses have been consistently successful.5
Despite recommendations and documented safety, HPV vaccine coverage among PLWH varies widely and is often below that of the general population, depending on healthcare system integration, provider engagement, and systematic implementation.5 Identified barriers include low perceived risk, vaccine hesitancy, lack of systematic clinician offering, and organizational issues such as incomplete referral pathways.5,7
Given these substantial gaps in HPV vaccination coverage and documented barriers to vaccination in PLWH, this study aimed to: 1) measure current HPV vaccination coverage in a tertiary HIV care cohort; 2) identify demographic and clinical determinants of vaccination completion; and 3) quantify system-level missed opportunities in the referral-to-vaccination pathway.
MethodWe conducted an observational, retrospective cohort study at the infectious diseases unit of Hospital Clínico Universitario de San Juan, Alicante, Spain, which followed 824 adult PLWH as of October 8th, 2024. Inclusion criteria were age ≥18 years, chronic HIV infection, and initial clinical care between 2009 and 2024, while exclusions comprised diagnosis before 2009, care at other hospitals, pregnancy, or preventive medicine service (PMS) referrals from 2025. This temporal window was selected to: 1) ensure sufficient historical data for retrospective ascertainment of vaccination status; 2) align with the period of systematic vaccination documentation in our electronic medical records; and 3) include patients with potential long-term follow-up data for referral and attendance tracking.
Of the 824 adults living with HIV followed at the unit, 212 met the eligibility criteria. After excluding 14 individuals due to incomplete data on key vaccination variables, the analytical sample comprised 198 patients.
Secondary data collected included basic patient information (such as age, sex, and nationality), clinical details (like treatment type, immune status, and time since diagnosis), and the source of their referral to the infectious diseases unit. Vaccination status was classified as complete, partial, not vaccinated, or unknown when records were lacking. Cases with unclear vaccination status due to missing information were excluded from the final analysis to ensure accuracy. Records of referrals and attendance at the PMS were monitored to assess whether patients continued with their follow-up. HPV vaccination status was categorized as complete (three doses), partial (one or two doses), unvaccinated, or not assessable when documentation was insufficient. Patients with not assessable vaccination status were excluded from the multivariable regression analyses, ensuring consistency between the total sample, the descriptive categories, and the analytical sample.
Loss to follow-up was defined as patients referred to PMS but had no subsequent record of HPV vaccination or follow-up visits in the hospital electronic medical records, suggesting non-attendance after referral. For vaccination status, these patients were classified as not vaccinated. In addition, loss to follow-up was included as a separate binary variable (yes/no) to explore its association with completion of the HPV vaccination schedule.
Data were extracted from a pseudonymized database of patients to minimize identification risk. Descriptive statistics summarized qualitative data as frequencies and percentages, and quantitative data as medians/interquartile range or means/standard deviation depending on distribution. Statistical tests were effectively use, a significance level set at p <0.05. Variables with p <0.20 in univariable analyses and clinical relevance determined inclusion in the multivariable logistic regression model; viral load was excluded due to collinearity with CD4 count. The final model adjusted for age, biological sex, CD4 nadir, time since HIV diagnosis, and time in study (continuous, years from HIV follow-up initiation). The data were analysed using version 29.0 of the SPSS software.
Ethics approval was granted by CEI of Hospital Clínico Universitario de San Juan, with informed consent waived for anonymized retrospective data. Procedures complied with the Declaration of Helsinki and data protection laws.
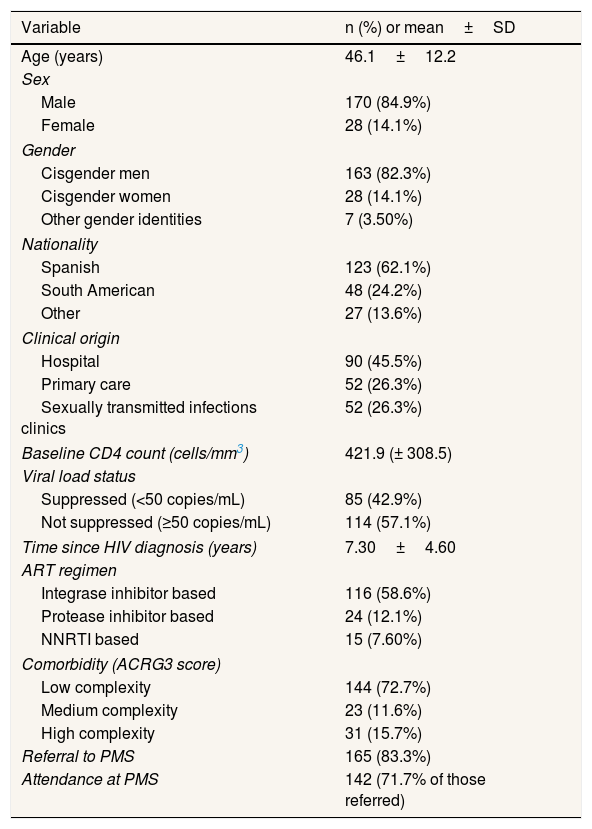
ResultsData collection from January 30 to March 3, 2025, yielded 198 patients after screening 212 records. Table 1 shows main cohort characteristics. Documentation showed that 83.3% were referred to PMS; of these, 71.7% attended, and 10.6% did not (excluding two individuals with appointments still pending). Among the 33 patients not referred to PMS (16.7%), reasons for non-referral included: clinician omission or lack of documented referral request (n=18, 54.5%), advanced clinical status considered a contraindication (n=7, 21.2%), transfer to another hospital during the study period (n=4, 12.1%), and explicit patient refusal or non-response to recommendation (n=4, 12.1%).
Sociodemographic and clinical characteristics of the sample (n=198).
| Variable | n (%) or mean±SD |
|---|---|
| Age (years) | 46.1±12.2 |
| Sex | |
| Male | 170 (84.9%) |
| Female | 28 (14.1%) |
| Gender | |
| Cisgender men | 163 (82.3%) |
| Cisgender women | 28 (14.1%) |
| Other gender identities | 7 (3.50%) |
| Nationality | |
| Spanish | 123 (62.1%) |
| South American | 48 (24.2%) |
| Other | 27 (13.6%) |
| Clinical origin | |
| Hospital | 90 (45.5%) |
| Primary care | 52 (26.3%) |
| Sexually transmitted infections clinics | 52 (26.3%) |
| Baseline CD4 count (cells/mm3) | 421.9 (± 308.5) |
| Viral load status | |
| Suppressed (<50 copies/mL) | 85 (42.9%) |
| Not suppressed (≥50 copies/mL) | 114 (57.1%) |
| Time since HIV diagnosis (years) | 7.30±4.60 |
| ART regimen | |
| Integrase inhibitor based | 116 (58.6%) |
| Protease inhibitor based | 24 (12.1%) |
| NNRTI based | 15 (7.60%) |
| Comorbidity (ACRG3 score) | |
| Low complexity | 144 (72.7%) |
| Medium complexity | 23 (11.6%) |
| High complexity | 31 (15.7%) |
| Referral to PMS | 165 (83.3%) |
| Attendance at PMS | 142 (71.7% of those referred) |
ACRG3: Adjusted Clinical Groups version 3; ART: antiretroviral therapy; HIV: human immunodeficiency virus; NNRTI: non-nucleoside reverse transcriptase inhibitors; PMS: preventive medicine service; SD: standard deviation.
Of the 198 patients, 39 (19.7%) were fully vaccinated, 21 (10.6%) had partially completed vaccination, 113 (57.1%) were unvaccinated, and 25 (12.6%) had insufficient documentation to assess HPV vaccination status. Among the subgroup aged ≤45 years (n=99), 28 (28.3%) had completed the full HPV vaccination series, 11 (11.1%) had received a partial vaccination, and 60 (60.6%) were unvaccinated. Stratified analysis by time since diagnosis showed that patients with longer HIV disease duration (≥5 years since diagnosis, n=145) had higher documented PMS referral rates (88.3%) compared to those with <5 years (n=53, 73.6%; p=0.03) and higher PMS attendance (75.2% vs. 63.5%; p=0.09).
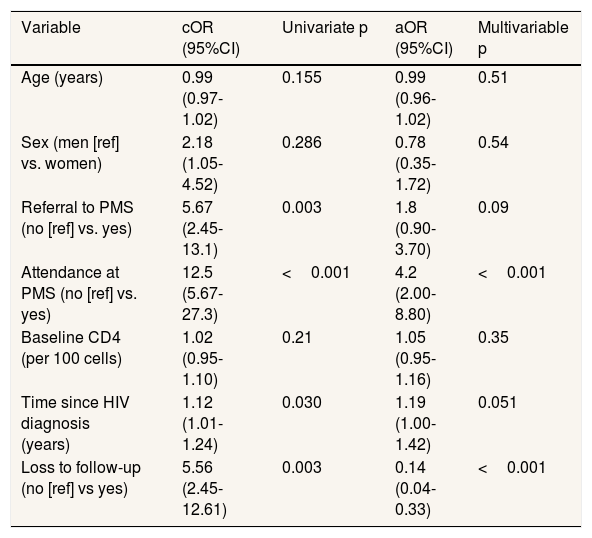
Table 2 presents the univariate and multivariable analyses of factors associated with completion of HPV vaccination. In univariate analysis all sociodemographic variables examined (age, sex, nationality, clinical origin) showed no significant association with vaccination completion (p>0.05 for all). Among clinical variables, baseline CD4 count (p=0.21) and viral load category (p=0.23) were not significantly associated with completion. Referral to PMS (p=0.003) and attendance at PMS (p=0.001) were significantly associated with vaccination completion and were selected for the multivariable model along with age and sex as potential confounders. Because CD4 count and viral load were correlated (r=0.68), only CD4 count was included in the multivariable model to prevent issues with multicollinearity.
Univariate and multivariate analysis of factors associated with complete human papillomavirus vaccination (n=198).
| Variable | cOR (95%CI) | Univariate p | aOR (95%CI) | Multivariable p |
|---|---|---|---|---|
| Age (years) | 0.99 (0.97-1.02) | 0.155 | 0.99 (0.96-1.02) | 0.51 |
| Sex (men [ref] vs. women) | 2.18 (1.05-4.52) | 0.286 | 0.78 (0.35-1.72) | 0.54 |
| Referral to PMS (no [ref] vs. yes) | 5.67 (2.45-13.1) | 0.003 | 1.8 (0.90-3.70) | 0.09 |
| Attendance at PMS (no [ref] vs. yes) | 12.5 (5.67-27.3) | <0.001 | 4.2 (2.00-8.80) | <0.001 |
| Baseline CD4 (per 100 cells) | 1.02 (0.95-1.10) | 0.21 | 1.05 (0.95-1.16) | 0.35 |
| Time since HIV diagnosis (years) | 1.12 (1.01-1.24) | 0.030 | 1.19 (1.00-1.42) | 0.051 |
| Loss to follow-up (no [ref] vs yes) | 5.56 (2.45-12.61) | 0.003 | 0.14 (0.04-0.33) | <0.001 |
aOR: adjusted odds ratio; 95%CI: 95% confidence interval; cOR: crude odds ratio; HIV: human immunodeficiency virus; PMS: preventive medicine service.
The results of the multivariable logistic regression model, adjusted for age, biological sex, CD4 nadir, time since HIV diagnosis, and time in study, show that attendance at PMS remained strongly associated with higher odds of vaccination completion (adjusted odds ratio [aOR]: 4.20; 95% confidence interval [95%CI]: 2.00-8.80; p <0.001). Similarly, longer time since HIV diagnosis was marginally associated with increased vaccination likelihood (aOR: 1.19; 95%CI: 1.00–1.42; p=0.051). Patients lost to follow-up had markedly lower odds of completing HPV vaccination (aOR: 0.14; 95%CI: 0.04–0.33; p=0.001), underscoring the impact of disrupted follow-up on preventive care. Further adjustment for time in the study did not materially change the results.
DiscussionMissed vaccination opportunities stemmed from lack of referral (16.7%) mostly due to clinician omission. Among referred patients, missed opportunities were due to non-attendance and incomplete scheduling (only 71.7% attendance). Patient refusal and prior vaccination accounted for fewer cases.
HPV vaccination completion in this cohort (19.7%) remains well below World Health Organization aspirational targets (≥90% coverage) for preventive vaccines and lower than documented coverage in the general Spanish population (estimated 60-75% in adolescents per Spanish vaccination registry data). Coverage in other PLWH cohorts varies widely (range: 5.60-90%) depending on healthcare system integration, intensity of provider engagement, and systematic vaccination program implementation.8,9
The main actionable determinant of vaccine completion was failure in the referral–attendance pathway. Attendance at PMS post-referral was the strongest independent predictor (aOR: 4.20), highlighting organizational and healthcare system barriers, including failure to refer and patient retention issues, as critical missed opportunities.
These findings align with Birk et al.,10 who found frequent clinical engagement and integrated vaccination services in an urban HIV clinic increased adherence. Prior literature emphasizes organizational and process barriers like incomplete clinician offering and referral breakdowns as main drivers of low vaccination among PLWH.8–10. Ringshall et al.11 stressed the importance of clinician training and health promotion in overcoming barriers.
Missed opportunities arise from two mechanisms: omission at care delivery (failure to refer/offer vaccination) and attrition after referral (non-attendance or incomplete scheduling).
Longer time since HIV diagnosis was associated with higher rates of documented PMS referral and attendance, suggesting that sustained engagement with longitudinal HIV care creates increased opportunities for preventive service integration. Conversely, recent diagnoses (<5 years) showed lower referral documentation, reflecting competing priorities during acute care or less integrated preventive services during patient stabilization phases. Loss to follow-up was a strong negative predictor of vaccination, indicating challenges in engaging vulnerable patients. Lack of significant association between socio-demographic or clinical factors (sex, baseline CD4, viral load) and vaccination completion underscores healthcare delivery roles over patient characteristics.
Interventions focusing on referral–attendance gaps, provider recommendation behaviour, and beliefs are most promising. Koskan et al.12 emphasized ongoing provider education to boost clinician confidence and normalize vaccination recommendations among all eligible HIV-positive adults, especially with expanded age criteria. Evidence-based strategies include: 1) provider education and normalization of vaccination recommendations; 2) automated or triggered referral systems; 3) point-of-care vaccination where feasible; 4) electronic reminder systems and SMS/email prompts; and 5) active patient navigation and re-outreach for lost-to-follow-up individuals.
Strengths of this study include random sampling, robust linkage of referral/attendance data, and multivariable modelling to isolate predictors. Limitations are retrospective design, single-center setting, reliance on routine clinical data susceptible to bias, lack of psychosocial/health-belief data, and limited subgroup analysis power limiting generalizability.
Bridging identified gaps requires practical, scalable system-level changes in tertiary care. Emphasis on automated referrals, point-of-care vaccination, electronic reminders, and focused retention efforts for patients with lost follow-up offers the clearest path to boost HPV vaccination in PLWH.
ConclusionsHPV vaccination coverage among PLWH remains suboptimal, with under one-third of those aged ≤45 completing vaccination. Attendance post-PMS referral is the strongest predictor of completion, highlighting systemic barriers such as referral failure and poor patient retention. Continuity of care and repeated clinician contact foster vaccination opportunities, while lost-to-follow-up patients constitute a vulnerable subgroup with low uptake. Addressing these via automated referrals, provider education, integrated vaccination delivery, and reminder systems is critical to reduce missed opportunities and mitigate HPV-related cancer risk in this population.
Availability of databases and material for replicationThe datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request. Materials and protocols used in this research can also be provided to facilitate replication of the results.
People living with HIV (PLWH) have increased risk of persistent HPV infection and related cancers including cervical, anal, and oropharyngeal cancers. In Spain, HPV vaccination is advised for PLWH up to age 26, according to GESIDA-SEIMC and the Ministry of Health (2018). According to EACS guidelines, people aged forty-five or younger may receive the vaccine if their personal risk warrants it. Vaccination is safe and immunogenic in PLWH, though coverage remains suboptimal compared to general population due to barriers like low perceived risk and healthcare system challenges.
What does this study add to the literature?This study provides updated HPV vaccination coverage in a Spanish tertiary care cohort of PLWH, showing less than a third of eligible patients’ complete vaccination. It identifies attendance at PMS as the strongest independent predictor of vaccination completion (aOR: 4.2; 95%CI: 2.0-8.8; p <0.001), while documented referral alone was not independently significant (aOR: 1.8; 95%CI: 0.9-3.7; p=0.09). These findings highlight that the referral-to-attendance gap represents the critical juncture for missed opportunities, with 28.3% of referred patients failing to attend PMS.
What are the implications of the results?Results underscore the need for system-level interventions including automated referrals, provider education, point-of-care vaccination, and patient reminder systems to increase vaccine uptake. Addressing these gaps can significantly reduce missed vaccination opportunities and HPV-related cancer risk in PLWH.
Alberto Lana.
Transparency declarationThe corresponding author, on behalf of the other authors guarantee the accuracy, transparency and honesty of the data and information contained in the study, that no relevant information has been omitted and that all discrepancies between authors have been adequately resolved and described.
Authorship contributionsS. Ivorra-Gómez: conceptualization, data analysis, drafting and critical review. F. Jover-Díaz: conceptualization, data analysis, study design, supervision, drafting and critical review and final approval of the manuscript. T. De Gea Velázquez-de Castro: data acquisition, methodology, and interpretation of results. E. Delgado-Sánchez: data curation, visualization, and revision of the final draft. J. Peris-García: statistical analysis, validation, and manuscript editing.
FundingNone.
Conflicts of interestNone.