The 1st International Conference on Safety and Public Health
Más datosTo describe the presence of bacteria at the seller's hand of meatball vendors and cart meatball bowls.
MethodThis is a descriptive study with an observational approach. Thirty mobile meatball vendors were recruited as participants. Therefore, the bacterias from bowls and hands were taken by swab sampling. The number of bacterias were obtained using total plate count method by means of nutrient agar. Bacteria types were identified using Gram staining method and bacteria assay.
ResultsThe hands and bowls swab samples were positive for Gram-positive and Gram-negative bacteria in all cases. These microorganisms include Acinetobacter calcoaceticus, Klebsiella sp., Alcaligenes faecalis, Enterobacter aglomereus, Pseudomonas aeruginosa, Enterobacter aerogenes, Enterobacter cloacae, and Staphylococcus saprophyticus, Serratia rubidaea, Yersinia enterocolitica, E. aerogenes, and Staphylococcus epidermidis.
ConclusionsSome types of bacterias were present in cart meatball bowls and the sellers’ hands. This phenomenon is affiliated with the emergence of food-borne diseases for the consumers.
Poor consideration for food safety is a major source of illness and has been affiliated with over 200 diseases, ranging from diarrhea to cancer.1 Sufficient amounts of safe and nutritious foodstuff are ingress to the sustenance of life and promotion of good health, while the unsafe variety possibly contains harmful bacteria, viruses, parasites, or chemical substances.2 In addition, bacteria and viruses are known to be the most common causes of food poisoning and other food-borne diseases and lots of people are at risk, with the possibility of losing life years.3 Moreover, some species of microorganisms have resulted in most illnesses, hospitalization, and death in the USA, comprising Salmonella, Norovirus, Campylobacter, Escherichia coli, Listeria, Clostridium perfringens.4
Food has a tendency to be toxic after contamination with pathogen bacteria. The microorganisms grow and proliferate during the storage process, while others naturally release toxins from fungi/plants and animals. The toxin-related bacteria include Salmonella, Shigella, Campylobacter, Listeria monocytogenes, Yersinia enterocolitis, and Staphylococcus aureus.5 One of the food-related incidence affecting fenugreek sprouts was reported in Germany, as a result of the E. coli enteropathogenic outbreak.6
Meatball is one of the popular street meat-based foods in Indonesia. Besides the taste and low-cost, this dumpling is also easily obtainable near houses, and sold by mobile hawkers, using a cart. Despite the already pass-through processing, meatballs are not entirely safe, especially from microbial contaminants from numerous sources. The high protein content acts as an ideal growth medium or substrate of pathogenic and other disease-causing organisms.7 In addition, high water content during processing has been affiliated with the accelerated growth, and result in a favorable pH for most enteric microorganisms.8
It is possible for products to be tainted by the meat handlers, as conveyors of microbes during the processes of producing, packing, and purchasing. Also, improper cooking and storage have been connected with the inception of food-borne illness,8 and cutlery in contact with the dish, including the bowl, potentially ensure contamination upon oral entry. The release of bacteria toxin into the body is known to trigger diseases.9,10
This study aims to analyze the presence of bacteria at the hands and cutlery of meatball cart vendors’ in Makassar city. The results are expected to contribute as information toward the improvement of vendor or awareness, in terms of safe handling practices and also to clarify the individuals’ potential role in food pathogen transmission.
MethodThis was a descriptive observational study, conducted in the three districts of Makassar City, including Makassar, Ujung Tanah, and Tamalanrea.
Sampling procedureThirty mobile meatball vendors were intercepted accidentally for this study. The bowls were randomly selected from the respective carts. The bacteria samples on the individuals’ hands and the bowls were obtained by swapping the surfaces with the swabbing tool. Therefore, the cotton swab from the bowl was dipped in sodium chloride (NaCl) (Merck, Darmstadt, Germany) solution for a minute and wiped into the inside of the bowl, from the middle to the left and to the right, three times. Also, the right hand was wiped from the thumb to the little finger and the palm, three times except for left-handed individuals. The swab was subsequently placed in a screw test tube containing NaCl, and sterilized with a Bunsen burner. This was then covered with sterile cotton, stored in a cool box, and transported to the laboratory for 24h incubation.
Bacteria analysisBacteria identification and biochemistry analysis involved similar procedure as observed with the previous study.11 The bacteria was assessed through total plate count (TPC) method, using nutrient agar (NA) (Oxoid Ltd, England) to calculate the total number of microorganisms, and MacConkey agar for identification. These medium inhibit the growth of Gram-positive bacteria, and facilitated the Gram-negative forms only.
Total plate countThe incubated sample and the NaCl were mixed using a vortex (Vibrofix VF1, IKA-Werk), and a total of 0.5ml was pipetted into the dilution reaction tube I (10−1) containing NaCl. Therefore, 0.5ml was pipetted from the dilution tube I to II (10−2) and from the II to III (10−3). Subsequently, 0.5ml sample was spiked into the sterilized petri dish (ANUMBRA) from III, followed by the addition of heated NA, which was then homogenized by swirling in an ‘8’ movement 12 times. The same process was conducted with MacConkey agar, and all petri dishes were incubated at 37°C for 24h, in an upside-down position. The bacteria colony was counted in colony-forming unit (CFU)cm−2.
Bacteria identificationEach bacterial colony smeared on the object glass (Sail Brand microscope slide 7101-7110-China) contained NaCl. This set up was fixed on the bunsen to dry, and then stained with Crystal violet (CI 42555-Merck, Germany) for 2–3min. Therefore, the surface was rinsed respectively with flowing water, lugol for 20s, flowing water, and 96% alcohol (OneMed, Indonesia) to ensure the color fades, before using flowing water, and Fuchsin alkali (CI 42510-Merck KGaA, Germany) for 2min. Finally, the smear was rinsed with clean flowing water, and drained on filter paper, followed by the observation of cell morphology using microscope (Nikon SE-PT New Module INT).
The Gram-positive bacteria tend to possess a thick mesh-like cell, formed by peptidoglycan (50–90%), conferring resistance toward the CV-Iodine complex, producing a purple pigmentation. Meanwhile, the gram-negatives comprise a relatively thinner layer of peptidoglycan, with cells appearing reddish or pink, which vanishes after rinsing with alcohol.12
Biochemistry assayDuring biochemical assay, nine agars were used as food source for bacteria. These microorganisms were stabbed and stroked into triple sugar iron agar (TSIA) (Merck KGaA, Germany) and inserted again into sulfide, indole, motility agar (SIM) (CM0435 SIM Medium Oxide LTD Basingstoke, England), before shaking on Methyl Red-Vogues Proskauer Agar (MRVP) (1.05712.0500 Microbiology MR-VP broth Merck KGaA, Germany). Furthermore, the sample was stroked again on citrate (CM0155 Simmons Citrate Agar Oxoid Ltd, Basingstoke, England) and urea agar (1.08492.0500 Urea Agar Merck KGaA, Germany). Therefore, the bacteria was collected again with an inoculating loop, before shaking sequentially on glucose (1.08337.0250 d(+)-Glucose Merck KGaA, Germany), lactose (7656.1000 Lactose-Monohydrate Merck KGaA, Germany), sucrose (7653 1000 Saccharose reinst Merck KGaA, Germany), and mannitol agar (1.05983.0250 d(−)-Mannitol Merck KGaA, Germany). Subsequently, the nine agar plates were arranged in a C-shelf test tube, incubated and observed for changes. These modifications were then matched with bacterial type tables, to determine the species present.
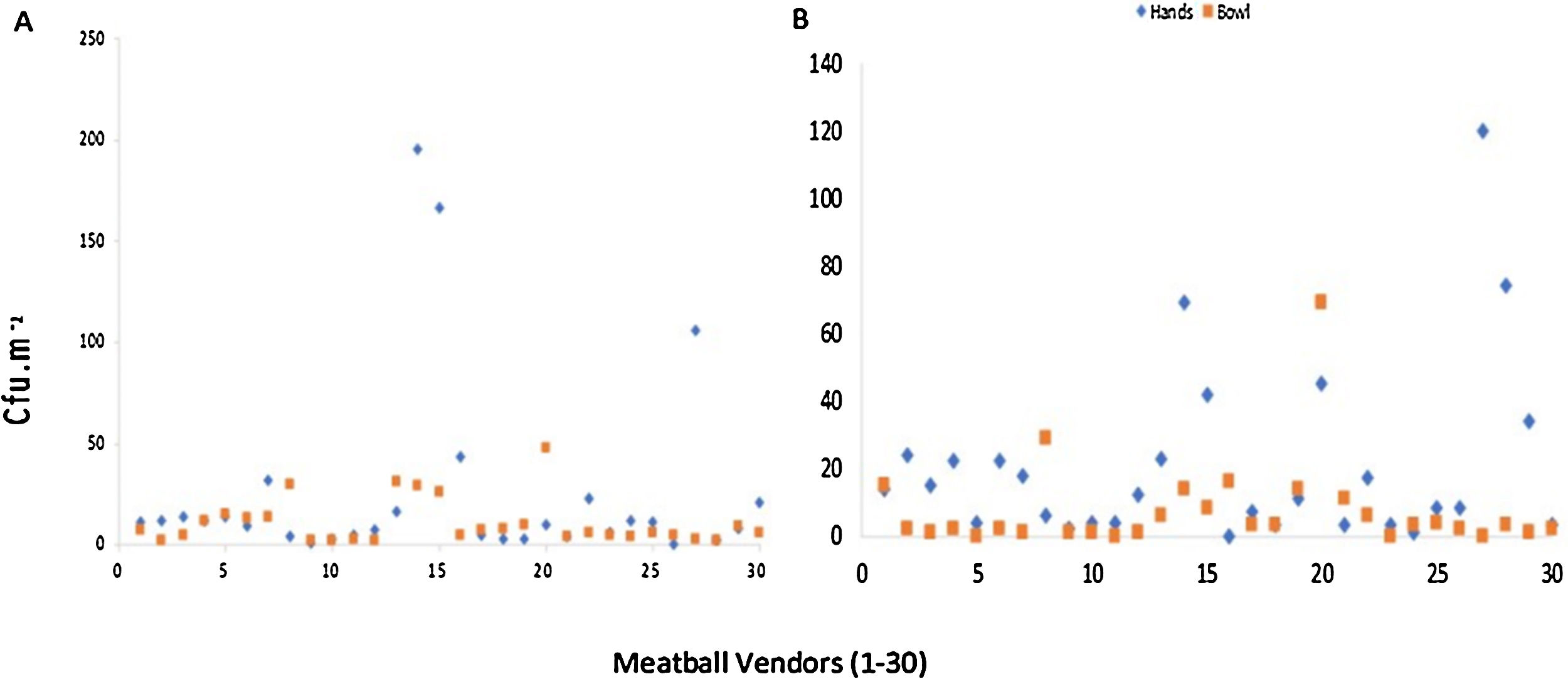
ResultsTotal plate countThe results show the detection of microbial contamination on both samples, and the growth colony in hands ranged from 0 to 1.95×102cfucm−2 in the NA medium and 0 to 1.20×102cfucm−2 on MacConkey medium (Fig. 1). There were six hands with TPC below 5cfucm−2 discovered on the NA medium, and nine on the MacConkay, while fewer had TPC of over 1.0×102cfucm−2. Moreover, the bowls possessed lesser TPC, with none exceeding 1.0×102cfucm−2. In addition, colony growth on the NA and MacConkey medium ranged from 2 to 4.8×101cfucm−2 and 0 to 6.9×101cfucm−2, respectively, while the TPC was below 5cfucm−2 for 9 and 16 bowls, using the corresponding medium.
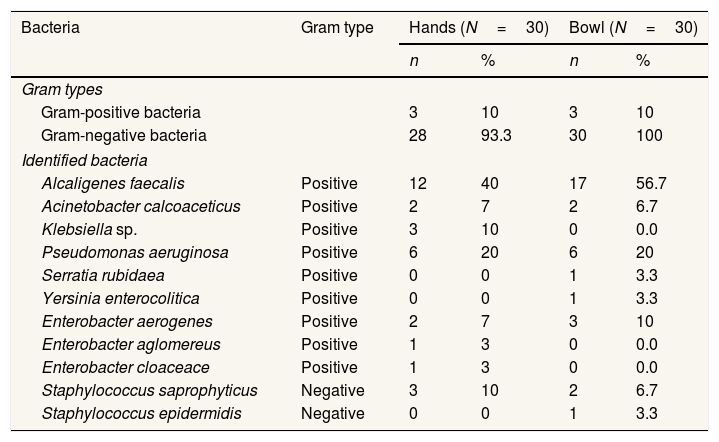
Bacteria typeBacteria were observed on all vendor hands and bowls, with results presented in Table 1. The gram-negative forms were dominant in both hands and bowls, and Alcaligenes faecalis was identified as the most significant colony at 40% and 56.5%, respectively.
Bacteria observed at hands and bowl of meatball cart vendors in Makassar City.
| Bacteria | Gram type | Hands (N=30) | Bowl (N=30) | ||
|---|---|---|---|---|---|
| n | % | n | % | ||
| Gram types | |||||
| Gram-positive bacteria | 3 | 10 | 3 | 10 | |
| Gram-negative bacteria | 28 | 93.3 | 30 | 100 | |
| Identified bacteria | |||||
| Alcaligenes faecalis | Positive | 12 | 40 | 17 | 56.7 |
| Acinetobacter calcoaceticus | Positive | 2 | 7 | 2 | 6.7 |
| Klebsiella sp. | Positive | 3 | 10 | 0 | 0.0 |
| Pseudomonas aeruginosa | Positive | 6 | 20 | 6 | 20 |
| Serratia rubidaea | Positive | 0 | 0 | 1 | 3.3 |
| Yersinia enterocolitica | Positive | 0 | 0 | 1 | 3.3 |
| Enterobacter aerogenes | Positive | 2 | 7 | 3 | 10 |
| Enterobacter aglomereus | Positive | 1 | 3 | 0 | 0.0 |
| Enterobacter cloaceace | Positive | 1 | 3 | 0 | 0.0 |
| Staphylococcus saprophyticus | Negative | 3 | 10 | 2 | 6.7 |
| Staphylococcus epidermidis | Negative | 0 | 0 | 1 | 3.3 |
In addition, a total of eight bacteria types were reported in each sample, including Enterobacter aerogenes and Staphylococcus saprophyticus.
DiscussionBacteria were positively identified in all samples evaluated in the current study, and the number of colonies grown varied based on the medium. In addition, some emerged at the NA, mainly positive and Gram-negative, instead of MacConkey, comprising only Gram-negative, while others grew on both. This phenomenon was due to the differences in microorganism characteristics.
A total of eight bacteria were identified at the hands. These microbes were potentially transmitted to the noodle (one of the additional ingredients when serving meatball), as the vendors and food handler tend to not practice hand washing before making contact with food. Therefore, it is important to maintain clean hands and avoid direct contact with food.13
The result from bowl evaluation showed the presence of eight bacteria as well. Furthermore, most microorganisms identified on hand were also in the bowl, although a few in the bowl were not discovered on the hand. The cleanliness of surfaces in contact ready-to-eat food is a critical point for safety,14 and it is impossible to eliminate all microbes without sterilization. This process is not feasible.
Alcaligenes faecalisA. faecalis was the most common bacterium recognized in hands and bowl, and is affiliated with the decomposition of food and any animal-based product, including fresh beef, poultry, fish, milk, eggs, including raw honey.15 Food contaminated with this species shows several changes, often detected through smell, taste, texture, as well as the formation and accumulation of mucus, gas, and liquid.16 This is an opportunistic bacterium implicated in urinary tract infection, keratitis bacterium, postoperative endophthalmitis, skin and soft tissue infection, bacteremia, meningitis, and wound infection. Research at an Elementary school canteen reported on the presence of A. faecalis, assumed emerge because of poor hygiene. This has led to hands and bowl contamination in dishwaters,17 therefore inducing decay in cold temperatures and promoting adverse health effects in humans. This species is responsible for bronchopneumonia, identified as a fatal disease, due to the high drug resistance in patients with dengue fever.18
Pseudomonas aeruginosaP. aeruginosa has been identified in numerous products with high organic content, including raw milk, minced beef, water, cheese, chicken breast, chicken wing, anchovy, and sardine.19 This study reported on the dominance of P. aeruginosa in bowls (7 samples), as the storage humidity supports the evolution and propagation.20 In contrast with other species, P. aeruginosa has an ability to grow at 37–42°C21 and act as a saprophyte, by naturally forming colonies in humans. Keskin and Ekmekci (2007) reported on the higher tendency for increased bacteria isolation during the summer, alongside an enhanced propensity to appeal at the soil, water, skin flora, as well as the general environment.20
Symptoms observed after infection in humans and animals include inflammation and blood poisoning (septicemia). Therefore, a rise in infection is observed following an obstruction of the host immune system, as shown in meningitis, wound and burns or surgical site infections, urinary tract infection, necrotic pneumonia, conjunctivitis, and cause fatal sepsis in infant, and also humans in weak condition.22
A total of 2700 amongst the approximately 32,600 infectious hospitalized patients in the United States die as a result of multidrug-resistant P. aeruginosa. This occurs after bacteria ingeniously evade the injected antibiotics, leading to unresponsive germs, and resistance development.23,24
Enterobacteriaceae familyEnterobacteriaceae is a group of bacteria, known to be the most challenging contaminant for raw and processed meat products worldwide.8 This is a large family of Gram-negative, rod-shaped, facultatively anaerobic bacteria, often used in food microbiology as indicator organisms for the assessment of overall products hygiene.25
There were three types of Enterobacter sp. identified in the vendors’ hands, including E. aglomereus, E. aerogenes, and E. cloacae, while the bowl only contained E. aerogenes. This is congruent with the report by Lues and Tonder (2007), where the Enterobacteriaceae family was confirmed in the food handlers’ hands (44%), with total viable counts (TVC) of between 5 and 1.8×101cfucm−2.26 Riskawati reported E. aerogenes as one of the 7 bacteria present in spoons and cups, as the dominant species.27
Generally, Enterobacter sp. lives in the human digestive tract, and also survives in clean water, wastewater, soil, plants, as well as food. The human isolates, including E. aerogenes and E. cloacae have been identified anywhere in the environment, and are collectively considered opportunistic pathogens. These species have also been implicated in nosocomial infection, urinary tract infection, surgical wound infection, and blood flow infection. In addition, the microorganisms tend to form colonies under the skin, respiratory tract, urinary tract, and digestive tract.28
This study is not in line with a research by Allam et al. (2016), where no food handlers’ possessed Enterobacteriaceae. In this current study, one participant had the highest Enterobacteriaceae on hands, also showed the most significant occurrence of TVC. A study performed by De Wit and Rombouts attributed this manifestation as a good indicator of personal and toilet hygiene, because contamination is possible regardless of toilet use.29 The numbers of Enterobacteriaceae diminishes through regular hand washing after defecation, and further lowers the fecal microorganisms.29
Staphylococcus saprophyticusS. saprophyticus is a gram-negative bacteria observed in both vendors’ hands and bowl. This is known to be a leading cause of urinary tract infection after E. coli, and is observed especially in sexually active young females. In addition, S. saprophyticus is classified as a normal flora in humans, situated at the perineum, rectum, urethra, cervix, and digestive tract. This is also generally placed in the gastrointestinal region of pig as well as cow, and is transferred to humans, through animal-based food. Furthermore, about half of all females experience this form of infection in their lifetime, and between 5% and 20% of non-hospitalized patients suffered from S. saprophyticus.30
Staphylococcus epidermidisS. epidermidis is a normal non-pathogenic flora of the skin, with the tendency to infect prostheses at orthopedic and cardiovascular.31 In addition, everyone has a potency risk of food-borne diseases, as the agents present in food have a tendency to be microorganisms or toxic compound. The dissemination of food-borne diseases is a broader public health issue, and also the primary cause of malnutrition in infants.32
The only Gram-positive group observed in bowls was S. epidermidis. This is a bacteria openly stored in the environment, and is recognized as a major cause of food poisoning, due to the intoxication ability. The effects of enterotoxin are perceived in the entire colon, and the severity level depends on the dosage, as well as patient endurance.31
Acinetobacter spp.Acinetobacter spp. was discovered in numerous sources, including water, soil, food, and arthropod. This bacterium is a leading cause of infection, and also possesses the ability to survive longer on dry surfaces, compared to other species. The difference observed in contrast with other gram-negative bacteria is the capacity to grow and develop in dry and humid areas, including healthy human skin. A study conducted in America attributed 63% of nosocomial infections in children hospitals to the presence of Acinetobacter spp. However, exposure to water and soil is perceived as a significant risk factor,33 thus a polluted environment is the probable cause of bacteria on hands and bowls. This is associated with the avoidance of gloves while peddling and the act of openly exposing bowls on storage.
Klebsiella sp.Klebsiella sp. consists of five species, including the pathogenic K.pneumoniae, and K.oxytoca, observed in high frequency as well as severity level. These species are encountered in the natural environment, including surface of drinking water, soil, vegetation, waste, industrial waste. Also, they have been strongly associated with the mucosal surface of humans, mammalian, birds, and reptiles. The K.pneumoniae sub-species are connected with chronic respiratory tract diseases and chronic nasal granulomatosis infection. These manifestations are familiar in tropical areas.34
ConclusionsAll tested samples, comprising the hands and bowls had a positive identification for bacteria. A total of three Gram-positive species were respectively recognized, while 27 and 30 Gram-negative were discovered, correspondingly. The bacteria present in the hands include Acinetobacter calcoaceticus, Klebsiella sp., A. faecalis, Enterobacter aglomereus, P. aeruginosa, E. aerogenes, Enterobacter cloacae, and S. saprophyticus while A. faecalis, A. calcoaceticus, P. aeruginosa, Serratia rubidaea, Yersinia enterocolitica, E. aerogenes, S. saprophyticus, and S. epidermidis were resident in the bowl. The presence of microorganisme in hands and food triggers the onset of food-borne illness to the consumers.
Ethics approval and consent to participateEthical approval was obtained from Ethic Committee, Faculty of Public Health, Hasanuddin University, using the letter number 2374/UN.4.14.8/TP.02.02/2019. Also, informed consent was obtained from the vendors prior to the collection of samples.
AcknowledgmentsThe authors are grateful to the laboratory technician for the assistance provided during analysis, and also to all the respondents for the participation and corporation.
Conflicts of interestThe authors declare no conflict of interest.
Peer-review under responsibility of the scientific committee of the 1st International Conference on Safety and Public Health (ICOS-PH 2020). Full-text and the content of it is under responsibility of authors of the article.