To assess the impact of introducing Xpert as a follow-on test after smear microscopy on the total number pulmonary TB notifications.
MethodGenexpert systems were installed in six departments across Guatemala, and Xpert was indicated as a follow-on test for people with smear-negative results. We analyzed notifications to national tuberculosis (TB) programmes (NTP) and the project's laboratory data to assess coverage of the intervention and case detection yield. Changes in quarterly TB notifications were analyzed using a simple pre/post comparison and a regression model controlling for secular notification trends. Using a mix of project and NTP data we estimated the theoretical yield of the intervention if testing coverage achieved 100%.
ResultsOver 18,000 smear-negative individuals were eligible for Xpert testing during the intervention period. Seven thousand, one hundred and ninety-three people (39.6% of those eligible) were tested on Xpert resulting in the detection of 199 people with smear-negative, Xpert positive results (2.8% positivity rate). In the year before testing began 1098 people with smear positive and 195 people with smear negative results were notified in the six intervention departments. During the intervention, smear-positive notification remained roughly stable (1090 individuals, 0.7%), but smear-negative notifications increased by 167 individuals (85.6%) to an all-time high of 362. After controlling for secular notifications trends over an eight-quarter pre-intervention period, combined pulmonary TB notifications (both smear positive and negative) were 19% higher than trend predictions. If Xpert testing coverage approached 100% of those eligible, we estimate there would have been a+41% increase in TB notifications.
ConclusionsWe measured a large gain in pulmonary notifications through the introduction of Xpert testing alone. This indicates a large number of people with TB in Guatemala are seeking health care and being tested, yet are not diagnosed or treated because they lack bacteriological confirmation. Wider use of more sensitive TB diagnostics and/or improvements in the number of people clinically diagnosed with TB have the potential to significantly increase TB notifications in this setting, and potentially in other settings where a low proportion of pulmonary notifications are clinically diagnosed.
Evaluar el impacto en la notificación de casos de tuberculosis pulmonar de la introducción de Xpert como prueba de continuación después del análisis microscópico.
MétodoSe instalaron sistemas Genexpert es seis departamentos de Guatemala y se indicó como prueba consecutiva en todos los resultados negativos en la baciloscopia microscópica. Se analizaron los datos del Programa Nacional y los del laboratorio del proyecto para medir la cobertura y la productividad en detección de casos. Las notificaciones trimestrales se compararon con los valores anteriores a la intervención y se adoptó un modelo de regresión para controlar por las tendencias temporales. Se estimó la contribución teórica de la intervención en términos de notificación si se obtuviera una cobertura del 100%.
ResultadosDurante el período de intervención, más de 18.000 personas con baciloscopia negativa fueron elegibles en los seis departamentos. El esputo de 7193 (36,9%) de ellos fue analizado también por Xpert y se detectaron 199 personas con baciloscopia negativa y Xpert positivo (positividad: 2,8%). En el año anterior a la intervención se notificaron 1098 casos de tuberculosis pulmonar y baciloscopia positiva, y 195 con baciloscopia negativa. Durante la intervención, la notificación de casos con baciloscopia positiva se mantuvo estable (1090 personas, 0,7%), pero las notificaciones con baciloscopia negativa, que incluía los casos con baciloscopia negativa y Xpert positivo, aumentó en 167 casos (85,6%), llegando a los 362 casos. Después de controlar por la tendencia temporal de notificación en los ocho trimestres anteriores, la notificación de tuberculosis pulmonar (con baciloscopia positiva o no) fue un 19% mayor que las predicciones de la tendencia. Si la cobertura de Xpert se acercase al 100%, se estima que se habría producido un incremento del 41% en las notificaciones.
ConclusionesSe identifica un importante aumento de las notificaciones de tuberculosis pulmonar solo con la introducción de Xpert. Ello indica que un número importante de personas con tuberculosis en Guatemala son atendidos por los servicios de salud y son sometidos a bacteriología microscópica, pero no son diagnosticados ni tratados porque no disponen de confirmación bacteriológica. La utilización de técnicas diagnósticas más sensibles o la mejora en el diagnóstico clínico tienen potencial para aumentar significativamente las notificaciones de tuberculosis pulmonar en esta zona y en cualquier otro lugar donde exista una proporción baja de diagnósticos clínicos no confirmados por microscopía.
While more than six million people with tuberculosis (TB) were treated and notified to national TB programs (NTP) in 2015, a further four million people remained undiagnosed and/or not notified. This poor case detection rate has been a persistently lagging indicator at a global-level for the past decade.1
When the Xpert MTB/RIF assay (Cepheid Sunnyvale, USA) was introduced, there were many expectations about how the test would impact the TB epidemic.2–4 Although use of the Xpert assay has steadily increased over the past five years,5 most of the testing outside South Africa has been focused on relatively small groups of people at higher risk for drug-resistant TB and only a handful of countries use Xpert as the first test for all people with suspected TB. The scale up of Xpert testing has increased the number of people identified with drug-resistant TB, as the Xpert assay rapidly provides a rifampicin resistance result and programs can be better link people with TB to appropriate care. However, controlled trials,6,7 programmatic evaluations8 and national data from South Africa,1 have shown that Xpert testing which replaces smear microscopy (e.g., no increase in testing volumes) does not improve the number of people initiated on drug-sensitive TB treatment, and in some cases, the scale up of Xpert testing has resulted in a decrease in TB treatment as clinicians may be less likely to diagnose TB clinically when faced with a negative Xpert result.
The overwhelming majority of Xpert cartridges are procured by African and Asian countries, with Brazil being the largest market in Latin and South America.9 Outside of Brazil, there are few examples of Xpert scale up from a programmatic context,10,11 possibly due to the region's lower burdens of human immunodeficiency virus (HIV) and drug-resistant TB and lower levels of external/donor funding which are often used to finance the more expensive test in other high TB burden settings.
Guatemala presents an interesting epidemiological setting in which to evaluate the introduction of Xpert testing, as its impact on TB treatment rates could be different from what has been observed in other high TB burden countries. Guatemala has a low HIV burden and a network of 290 health facilities equipped with quality-assured smear microscopy for a country of 15.5 million inhabitants. According to the World Health Organization's (WHO) 2016 TB figures, Guatemala detected and notified 80% (confidence interval: 64-100%) of its estimated incident TB burden.1 However, in the previous year the WHO estimated Guatemala's case detection rate to be just 34% (confidence interval: 31-39%), indicating the level of uncertainty in TB burden estimates for countries which have not conducted a recent TB prevalence survey.12 Other Central American countries have had consistently higher case detection rates (87% in Salvador, 82% in Honduras and 76% in Nicaragua).1 A very high proportion of Guatemala's new pulmonary adult TB notifications are smear-positive (85% in 2012) compared to other countries in the region (78%) and globally (56%). Given the reliance on smear microscopy despite its low sensitivity,13 the possible low case detection rate in Guatemala, and the low proportion of clinically diagnosed pulmonary TB, we designed an evaluation to assess the impact of introducing Xpert as a follow-on test after smear microscopy on the total number pulmonary TB notifications.
MethodStudy settingIn collaboration with the NTP, six departments with a population of 4.6 million people and covering approximately one third of the country's reporting BMUs (Basic Management Units) (Fig. 1) were identified as sites for installation of GeneXpert systems and the scale up of routine Xpert testing, based on the ability to centrally monitor project implementation and on the historical numbers of people treated for TB in each department. The city of Guatemala is divided into four sub-departments, of which two were included in the project's intervention area.
Introduction of Xpert testingThree 4-module and four 2-module GeneXpert machines were placed in six large reference hospitals in each department (Retalhuleu Department received two machines). A total of 319 healthcare workers from 155 districts including laboratory technicians, physicians and nurses were trained on the intervention testing algorithm, use of the machines, sample preparation, completion of forms and referring samples covering the intervention area's 107 health facilities.
Between April 1, 2015 and March 31, 2016, any person with a smear-negative result from any of the health centers within the six intervention departments was considered eligible to receive an Xpert test. The same specimen which was used for smear microscopy was processed for the Xpert test. If multiple specimens were available from the same person, the laboratory technician chose the best quality specimen for Xpert testing. Specimens were transported from peripheral laboratories and collection sites using locally available resources to allow for as much coverage as possible. Only existing health center staff were used in the study, and they were responsible for the collection, selection, registration, and testing of the specimens. Anyone with an Xpert-positive result was notified to the TB officer of the department and initiated on treatment at the respective facility. People with smear-negative, Xpert-positive TB were registered in the national information system as smear-negative in accordance with national guidelines at the time, with a note in the register explaining they were Xpert-positive so the impact of the intervention could be tracked.
Each GeneXpert system was connected to GxAlert software14 to allow for in real-time, centralized data monitoring by program coordinators. When Xpert-positive results were transmitted via the GxAlert system, the program team would review the laboratory and treatment registers and patient cards to ensure the proper registration in the national system, as well as in the study database.
Data collection and analysisProject test data were analyzed to produce descriptive statistics and assess testing yields. Aggregate diagnostic smear microscopy testing data were collected from the NTP starting from the year before the project began. We used these data to calculate Xpert testing coverage, defined as the number of people tested by Xpert divided by all people with smear-negative results.
To evaluate the effect of Xpert testing on TB notifications, eight quarters of historical TB notification data were collected from all six intervention departments. Changes in the TB notifications by type of TB were measured between the intervention period (1 April 2015 to 31 March 2016) and the year immediately prior (1 April 2014 to 31 March 2015), in line with a standard evaluation framework for intensified TB case finding initiatives.15,16 In addition, we fitted a linear regression trend line through all eight quarters of historical TB notification data by type of TB to compare trend-expected and actual notifications during the intervention period.
Since not all people with smear-negative results could be tested on Xpert due to cost restrictions and logistical limitations, we also modelled the theoretical impact of expanding Xpert testing coverage by assuming yields would be 30% lower than the actual yields found during the intervention. We also modelled the impact of using the new Xpert MTB/RIF Ultra assay.15,17
The study was approved by the NTP of Guatemala and since it included only programmatic data collected through the standard NTP registers with no personal identifiers, ethical clearance was not required.
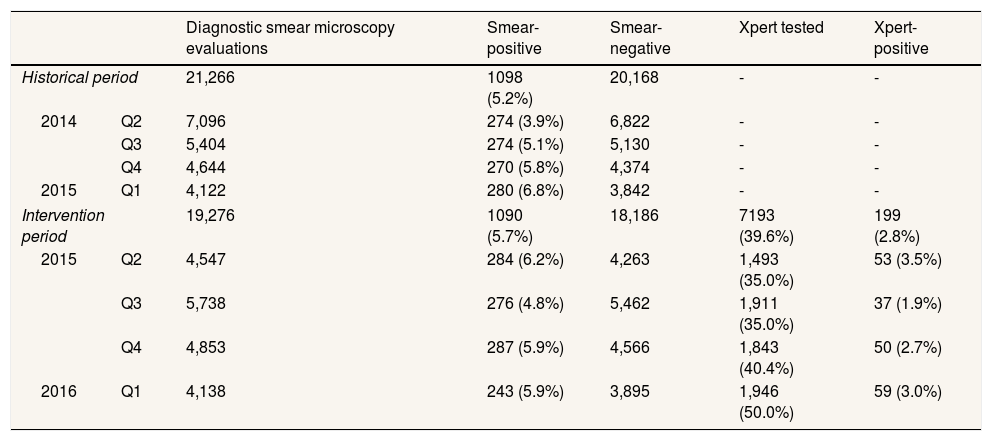
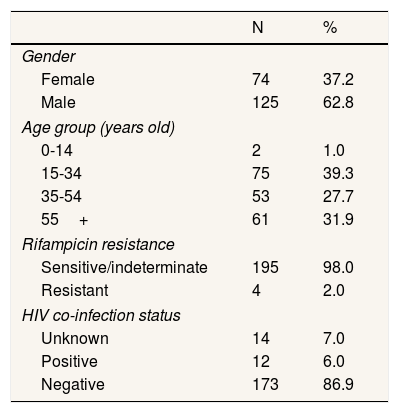
ResultsTable 1 shows a summary of the NTP's diagnostic smear microscopy testing rates and yields, as well as the coverage and yield of Xpert testing. The NTP laboratory data show that the number of diagnostic smear investigations declined by almost 10% year-on-year at the time Xpert testing was introduced. However, smear-positive TB yields remained stable (5.2% in year prior vs 5.7% during the intervention; p=0.03). During the intervention period, 19,276 people underwent a diagnostic smear microcopy evaluation in the six intervention departments. Of people who were smear tested, 1090 (5.7%) were diagnosed with smear-positive TB. 7193 people with smear-negative results were then tested on Xpert (39.6% of those who were eligible). Xpert coverage increased from 35.0% in the intervention's first quarter to 50.0% by the end of the evaluation. 199 people were detected with smear-negative, Xpert-positive TB (2.8% of those tested), and just 4 (2.0%) of these people had rifampicin-resistant TB. Table 2 shows the demographic details of Xpert-positive individuals. More males than females were diagnosed with Xpert-positive TB and the largest percentage of Xpert-positive results (39.0%) were among people aged 14-34 years. HIV results were not available for all Xpert-positive individuals, however just 12 (6.5%) had a documented HIV co-infection.
Diagnostic smear microscopy and Xpert MTB/RIF testing rates/coverage and yields.
| Diagnostic smear microscopy evaluations | Smear-positive | Smear-negative | Xpert tested | Xpert-positive | ||
|---|---|---|---|---|---|---|
| Historical period | 21,266 | 1098 (5.2%) | 20,168 | - | - | |
| 2014 | Q2 | 7,096 | 274 (3.9%) | 6,822 | - | - |
| Q3 | 5,404 | 274 (5.1%) | 5,130 | - | - | |
| Q4 | 4,644 | 270 (5.8%) | 4,374 | - | - | |
| 2015 | Q1 | 4,122 | 280 (6.8%) | 3,842 | - | - |
| Intervention period | 19,276 | 1090 (5.7%) | 18,186 | 7193 (39.6%) | 199 (2.8%) | |
| 2015 | Q2 | 4,547 | 284 (6.2%) | 4,263 | 1,493 (35.0%) | 53 (3.5%) |
| Q3 | 5,738 | 276 (4.8%) | 5,462 | 1,911 (35.0%) | 37 (1.9%) | |
| Q4 | 4,853 | 287 (5.9%) | 4,566 | 1,843 (40.4%) | 50 (2.7%) | |
| 2016 | Q1 | 4,138 | 243 (5.9%) | 3,895 | 1,946 (50.0%) | 59 (3.0%) |
Demographics details of Xpert-positive individuals.
| N | % | |
|---|---|---|
| Gender | ||
| Female | 74 | 37.2 |
| Male | 125 | 62.8 |
| Age group (years old) | ||
| 0-14 | 2 | 1.0 |
| 15-34 | 75 | 39.3 |
| 35-54 | 53 | 27.7 |
| 55+ | 61 | 31.9 |
| Rifampicin resistance | ||
| Sensitive/indeterminate | 195 | 98.0 |
| Resistant | 4 | 2.0 |
| HIV co-infection status | ||
| Unknown | 14 | 7.0 |
| Positive | 12 | 6.0 |
| Negative | 173 | 86.9 |
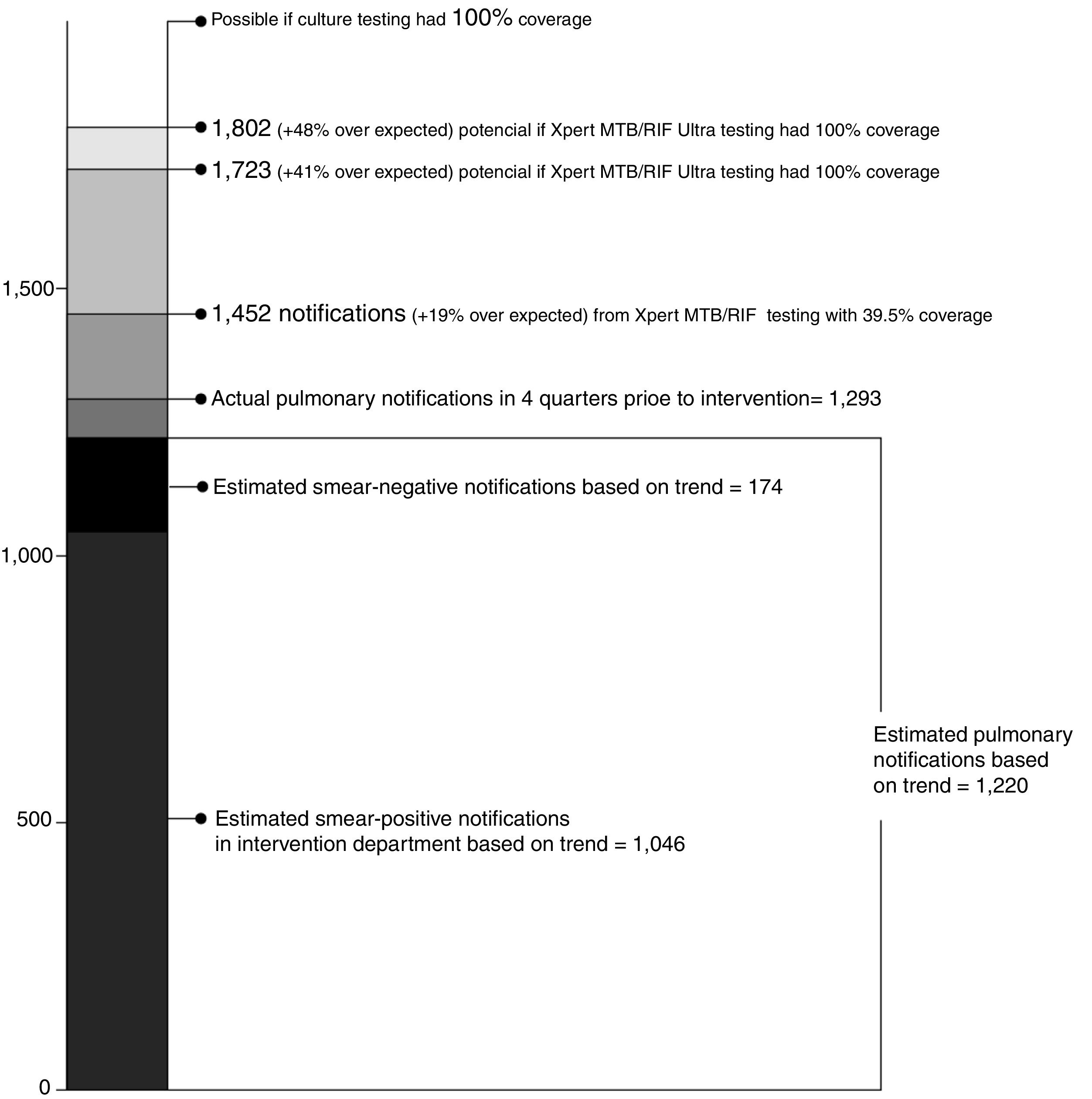
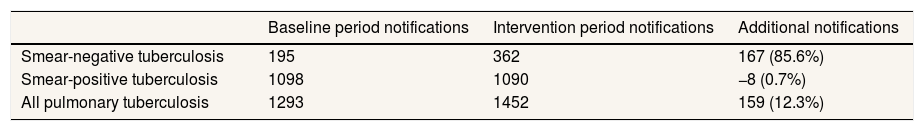
Table 3 shows how pulmonary TB notifications changed during the intervention period across the six intervention departments. During the four quarters immediately preceding introduction of Xpert, 195 people with smear-negative TB and 1098 people with smear-positive TB were notified (total of 1,293 people with pulmonary TB). During the four quarters when Xpert tested was introduced, 362 smear-negative individuals were notified (85.6%), while smear-positive enrolments remained stable. The gain in smear-negative notifications, driven by the increase bacteriological confirmation of TB from Xpert testing, increased total pulmonary notifications by 12.3%. After adjusting for secular notification trends in the pre-intervention quarters, smear-negative notifications were 108.0% above trend expectations and all pulmonary TB was 19.1% higher.
Changes in tuberculosis notifications across the six intervention departments.
| Baseline period notifications | Intervention period notifications | Additional notifications | |
|---|---|---|---|
| Smear-negative tuberculosis | 195 | 362 | 167 (85.6%) |
| Smear-positive tuberculosis | 1098 | 1090 | −8 (0.7%) |
| All pulmonary tuberculosis | 1293 | 1452 | 159 (12.3%) |
| Trend-expected notifications during the intervention period | Actual notifications during the intervention period | Additional notifications | |
|---|---|---|---|
| Smear-negative tuberculosis | 174 | 362 | 188 (108.0%) |
| Smear-positive tuberculosis | 1046 | 1090 | 44 (4.2%) |
| All pulmonary tuberculosis | 1220 | 1452 | 232 (19.1%) |
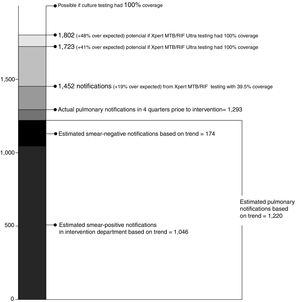
If Xpert testing coverage among people with smear-negative results reached 100%, rather than the 39.6% actually achieved, an additional 271 people with Xpert-positive TB could have possibly been identified (Fig. 2). If all of these people were linked to treatment, 1,723 people with pulmonary TB might have been notified across the six intervention departments —a 41.2% increase over trend-expected notifications. Given the superior sensitivity the Xpert MTB/RIF Ultra assay,18,19 an additional 350 people with Ultra-positive TB might have been identified —a 47.7% increase over trend expected notifications.
DiscussionThis programmatic evaluation of the introduction of Xpert testing suggests that a large number of people with TB who currently access health services in Guatemala are not being correctly diagnosed, resulting in missed opportunities for treatment, continued transmission of TB in communities and preventable deaths. These findings contrast with published studies from other high TB burden settings which have documented no increases (or even declines) in population-level TB treatment figures after the introduction of more sensitive TB diagnostic tools.6–8 We find that Xpert testing alone resulted in a 12.3% increase in pulmonary TB notifications (19.1% after controlling for secular notification trends), despite only achieving a testing coverage of 39.6%. The increases in pulmonary TB notifications directly correlates with the timing of Xpert testing introduction and the yield from Xpert testing.
Ultimately, our findings underscore the important role that clinical diagnosis must play in the TB response, especially when smear testing remains commonplace. Many groups are likely to be missed by smear microscopy given its low sensitivity, including people living with HIV,17,20 children18,21 and women.20,22 Although people with smear-positive TB are likely more infectious, people with smear-negative TB contribute to a substantial portion of transmission21,23,24 and more efforts must be placed on identifying these harder to diagnose people.
We believe the gains in pulmonary notifications measured in this evaluation are due in large part to low rates of clinical diagnosis in the period before the intervention and that these finding could possibly be replicated in other settings where clinical diagnosis is similarly limited. We measured gains in TB notifications with relatively low laboratory yields, and less than 40% testing coverage among our target population. Our results suggest that even greater gains could be achieved with higher testing coverage, perhaps up to a 41% increase in TB treatment. In addition, the new Xpert MTB/RIF Ultra test has an even higher sensitivity, particularly among people with smear-negative results15 and could increase gains even further (perhaps a 48% increase). Further studies are needed to document how new, highly sensitive TB diagnostics impact clinicians’ decisions on diagnosing TB in the absence of bacteriological confirmation.
Our findings also highlight the challenge TB programs face when developing interventions and investment plans based on WHO TB burden point estimates, which can fluctuate from year to year and come with wide confidence intervals when no direct measures of TB burden are available. We have conclusively showed that a substantial number of people with TB are currently being missed in Guatemala despite official statistics showing a case detection rate of 80%.
The use of GxAlert greatly improved the ability of program coordinators to monitor the intervention. Results were transmitted in real-time which enabled prompt follow-up with local facilities for positive and failed test results. This program was not the first to introduce Xpert testing in Guatemala, as there were a few GeneXpert machines used for drug resistance testing before this intervention. However, the importation of the machines and cartridges was not as easy as has been reported elsewhere.5,23,25 High importation taxes on the Xpert cartridges added 50% to the per test cost and the increased cost will hamper efforts to increase testing volumes and achieve universal drug susceptibility testing targets.24-27 While some studies have found Xpert to be cost savings or cost neutral,16,19,26,27 this will likely vary by setting and we have not attempted any costing analysis.
This was not a strictly controlled trial, but rather a programmatic evaluation and external factors may have influenced TB notifications, including a large health worker strike which contributed to the decline in diagnostic smear evaluations during the intervention period. However, our results reflect the realities of introducing and scaling up Xpert testing in Guatemala on a programmatic level. Guatemala is testing a considerable number of people for TB by microscopy and has a large network of laboratories. The yields from three smears under routine conditions are very low (about 5%), which is in stark contrast to the yields from passive case finding in other high TB burden settings.22 Similar to other studies where Xpert was used as a follow-on test after a negative smear result, the yields of Xpert were less than microscopy, but are still noteworthy since they were testing only those with smear-negative results.23
Xpert introduction usually does not increase the total TB case notification although it increases the notification of bacteriologically confirmed cases at the expense of notifying less clinically diagnosed ones.
What does this study add to the literature?The use of Xpert can increase the total TB case notification in areas, such as Guatemala, where the percentage of notified cases that are only clinically diagnosed over total notified cases is low.
Enrique Castro-Sánchez.
Transparency declarationThe corresponding author on behalf of the other authors guarantee the accuracy, transparency and honesty of the data and information contained in the study, that no relevant information has been omitted and that all discrepancies between authors have been adequately resolved and described.
Authorship contributionsAll authors have contributed in the conception and design of this work. MEG and SOF carried the weight of the data collection. Interpretation of results was carried out by all the authors in several work sessions. The first draft was prepared by JC, AJC, MEG and SOF and they and the rest of the authors reviewed it critically, approved the final version and are responsible for all its content.
FundingThe intervention on which this article is based was funded by the Wave 3 of TB REACH (STOP TB PARTNESHIP). TB REACH is funded by Global Affairs Canada.
Conflicts of interestNone.