To assess the monetary savings resulting from a pharmacist intervention on the appropriateness of prescribed drugs in community-dwelling polymedicated (≥8 drugs) elderly people (≥70 years).
MethodAn evaluation of pharmaceutical expenditure reduction was performed within a randomised, multicentre clinical trial. The study intervention consisted of a pharmacist evaluation of all drugs prescribed to each patient using the “Good Palliative-Geriatric Practice” algorithm and the “Screening Tool of Older Persons Prescriptions/Screening Tool to Alert doctors to Right Treatment” criteria (STOPP/START). The control group followed the routine standard of care. A time horizon of one year was considered and cost elements included human resources and drug expenditure.
Results490 patients (245 in each group) were analysed. Both groups experienced a decrease in drug expenditure 12 months after the study started, but this decrease was significantly higher in the intervention group than in the control group (−14.3% vs.−7.7%; p=0.041). Total annual drug expenditure decreased 233.75 €/patient (95% confidence interval [95%CI]: 169.83-297.67) in the intervention group and 169.40 €/patient (95%CI: 103.37-235.43) in the control group over a one-year period, indicating that 64.30 € would be the drug expenditure savings per patient a year attributable to the study intervention. The estimated return per Euro invested in the programme would be 2.38 € per patient a year on average.
ConclusionsThe study intervention is a cost-effective alternative to standard care that could generate a positive return of investment.
Evaluar los ahorros monetarios resultantes de la intervención de un farmacéutico orientada a mejorar la adecuación de los fármacos prescritos en ancianos (≥70 años) polimedicados (≥8 medicamentos) de la comunidad.
MétodoSe evaluó la reducción del gasto farmacéutico en el marco de un ensayo clínico aleatorizado y multicéntrico. La intervención del estudio consistió en una evaluación de todos los fármacos prescritos a cada paciente utilizando el algoritmo Good Palliative-Geriatric Practice y los criterios Screening Tool of Older Persons Prescriptions/Screening Tool to Alert doctors to Right Treatment (STOPP/START). El grupo control siguió la práctica clínica habitual. Se consideró un horizonte temporal de un año y los elementos de costes incluyeron los recursos humanos y el gasto en medicamentos.
ResultadosSe analizaron 490 pacientes (245 por grupo). La disminución del gasto farmacéutico a los 12 meses fue significativamente mayor en el grupo de intervención que en el grupo control (−14,3% vs.−7,7%; p=0,041). El gasto anual en medicamentos disminuyó 233,75 € por paciente (intervalo de confianza del 95% [IC95%]: 169,83-297,67) en el grupo de intervención y 169,40 € por paciente (IC95%: 103,37-235,43) en el grupo control, indicando un ahorro farmacéutico de 64,30 € por paciente/año atribuible a la intervención del estudio. Se ha estimado un retorno de 2,38 € por cada euro invertido en el programa.
ConclusionesLa intervención en estudio es una alternativa rentable a la atención estándar, que podría generar un retorno positivo de la inversión.
Population ageing has led to an increase in the prevalence of chronic diseases and in the use of health resources including medication. People over 65 years, which have a high prevalence of chronic diseases and are often treated with multiple drugs,1 represent 17% of the current Spanish population and are responsible of 70% of pharmaceutical expenditure.2 Moreover, in the last years, pharmaceutical expenditure has growth much more than the gross domestic product in most European countries, threatening the sustainability of public health care systems.3 On the other hand, potentially inappropriate prescribing (PIP) comprises a number of suboptimal prescribing practices, including inappropriate dose or duration of medication, drug-drug interactions, drug-disease interactions, and use of medications that have a significant risk of an adverse drug event.4,5 PIP has been found to be frequent in elderly population and associated with morbidity, adverse drug events, hospitalizations, and health care expenditures.6,7 For this reason, strategies aimed to improve the quality and safety of prescription in the elderly population can generate substantial health and economic benefits.8,9 Several criteria and algorithms have been developed to reduce PIP. One of the most used criteria is the Screening Tool of Older Persons Prescriptions/Screening Tool to Alert doctors to Right Treatment (STOPP/START).10 Likewise, an algorithm used to enable more rational and appropriate use of medication in elderly people is the Good Palliative-Geriatric Practice (GP-GP).11 Some authors have assessed the safety and effectiveness of interventions using these tools. Improvements in drug appropriateness and reduction in number of medications have been reported.12,13 Additionally, strategies including a clinical pharmacist can help decrease PIP and optimize patient therapy, resulting in better clinical outcomes.14 However, limited evidence exists about economic evaluations of these interventions.15,16 The objective of the present study was to assess the economic impact in drug expenditure of a pharmacist intervention on prescriptions to community-dwelling polymedicated elderly people.
MethodClinical trial designA randomized, open-label, multicenter, parallel-arm clinical trial was conducted in seven primary care centers in the city of Mataró and Argentona (Barcelona, Spain) (122,905 and 11,718 inhabitants respectively, with 13,290 and 1,194 aged 70 years and over, respectively) to assess the effect of a pharmacist intervention on the appropriateness of prescribed drugs. Details of this study were published elsewhere.17 In brief, the study population included a randomly selected sample of community-dwelling (non-institutionalized) elderly people aged 70 years and older, receiving eight or more drugs. Recruitment took place from February and May 2012 and participants were pre-selected from the primary care database and randomized with allocated concealment to one of the two study arms. The intervention, which took place no more than a month after the recruitment visit, included a trained and experienced clinical pharmacist evaluating all drugs prescribed to each patient using the GP-GP algorithm and basing their decision about appropriateness on the STOPP/START criteria. The pharmacist discussed recommendations for each drug with the patient's physician on a face-to-face visit in order to come up with a final set of recommendations (stop, start or change medication or dosage). Finally, these recommendations were discussed with the patient, and a final decision was agreed by physicians and their patients in a face-to-face routine visit. A safety control visit was planned one month later. All changes in prescribed medication were registered in the electronic clinical notes and in the study's record form. The control group received usual clinical practice. Overall, 503 patients were recruited; 251 in the control group and 252 in the intervention group, in which 2709 drugs were evaluated. The study protocol was approved by the local ethical committee (CEIC 05/12) and all participants gave their consent by writing before inclusion.
Cost elements consideredWe aimed to assess the reduction in pharmaceutical expenditure alongside the clinical trial due to the pharmacist intervention in primary care in relation to the routine clinical practice (standard of care by the general practitioner), following the ISPOR Good Research Practices report for reporting economic evaluation alongside clinical trials.18 We have considered a time horizon of one year following intervention.
Cost elements considered in the study included particularly the use human resources and consumption of drugs. In relation to human resources, it was estimated that the study intervention required a mean of 30minutes of a pharmacist per patient (drug evaluation and discussion with physician) and 20minutes of a physician per patient (discussion with pharmacist and an additional visit with the patient). Monetary valuation of time was possible using salary data available through the 2012 collective labor agreement (Catalan Health Service). The cost per hour, for both physicians and pharmacists, was 32.44 €/hour which included social security contributions but not structural costs. Drug prices, number of dispensed prescriptions, number of “generic drugs” and number of “new drugs” were obtained from the administrative pharmacy database of the Catalan Health Service. “New drugs” are considered drugs commercialized over the last five years and qualified with category C (most suitable therapeutic alternatives exist) or D (alternative therapeutic comparative information is not conclusive) by the Catalan Health Service. Only dispensed medication costs were considered, so real cost in drug expenditure was contemplated. The return of investment of the study intervention has been calculated dividing the savings in drug expenditure by the cost of the intervention.
Data analysisA sensitivity analysis was performed based on three theoretical scenarios, the first being the basal scenario (30minutes pharmacist and 20minutes physician time), the second being a rather more optimistic one (20minutes pharmacist and 15minutes physician), and a third one labeled as conservative scenario (40minutes pharmacist and 30minutes physician per patient). Among recruited patients, 13 (2.58%) died during the 12 months follow-up (six in the control group and seven in the intervention group), and were excluded from the cost analysis. The number of dispensed drugs in the 12 months before the intervention was also obtained using the same data sources. The difference between study groups in drug consumption 12 months before and 12 months after the intervention was also computed. Continuous variables (total drug expenditure 12 months after intervention, incremental drug expenditure 12 months before versus after intervention, total number of recipes 12 months after intervention and incremental number of recipes 12 months before versus after intervention) were described using means or medians (in case of non-normal distribution) and standard deviations. Basal and follow up comparisons between groups were tested by the t-test (for variables with normal distribution) or the Mann Whitney U test (for variables without normal distribution). Statistical significance was established at a p value<0.05.
To estimate the sample size, the main outcome variable was the pharmaceutical expenditure 1 year before and 1 year after the study intervention (paired data). For an alpha risk of 0.05 and a beta risk of 0.2 (two-tailed tests), it was estimated that 81 subjects were required to detect a difference equal to or greater than 250 € in pharmaceutical expenditure, assuming a standard deviation (SD) of 800 €.
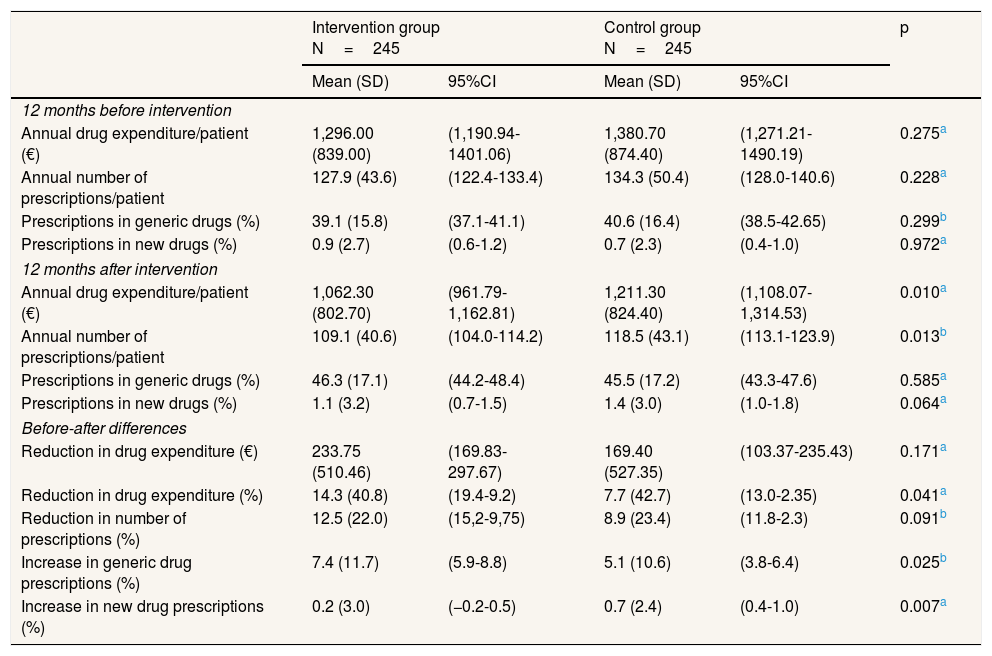
ResultsAfter excluding patients who died (n=13), 245 patients were recruited to control group (78.7 years, SD: 5.5; 57.9% women) and 245 to intervention group (79.1 years, SD: 5.4; 61.6% women). No statistically significant differences were observed between the two groups in the number of drug prescriptions and drug expenditure during the 12 months before pharmacist intervention, indicating that both groups were originally comparable. We observed, however, statistically significant differences in drug expenditure and in the number of prescriptions during the 12 months after intervention. As shown in Table 1, although both groups experienced a decrease in drug expenditure 12 months after the study started, this decrease was significantly higher in the intervention group than in the control group. Similarly, the rise in generic drug prescriptions was significantly greater in the intervention group. Table 1 also reports on total cost and total number of prescriptions before and after the pharmacist intervention. It shows a significant reduction for both groups in total costs, in the total number of prescriptions, and a significant increase in both groups in the percentage of generic drugs prescribed. We also observed a significant increase in the number of new drug prescribed in the control group. Total annual drug expenditure in the intervention group decreased from 317,520.00 € (pre-intervention) to 260,263.00 € (post-intervention), representing 233.75 € (95% confidence interval [95%CI]: 169.83-297.67) saved per patient (1,296.00 €/patient pre vs. 1,062.30 €/patient post). Similarly, total annual drug expenditure in the control group decreased from 338,271.00 € to 296,768.00 €, which results in 169.40 € (95%CI: 103.37-235.43) saved per patient over one-year period (1,380.70 €/patient pre vs. 1,211.30 €/patient post). The resulting incremental drug expenditure of standard care over pharmacist intervention is 64.30 € per patient each year. In other words, 64.30 € would be the cost savings per patient a year attributable to the study intervention.
Prescription patterns and associated annual costs/expenditures per patient before and after the pharmacist intervention.
| Intervention group N=245 | Control group N=245 | p | |||
|---|---|---|---|---|---|
| Mean (SD) | 95%CI | Mean (SD) | 95%CI | ||
| 12 months before intervention | |||||
| Annual drug expenditure/patient (€) | 1,296.00 (839.00) | (1,190.94-1401.06) | 1,380.70 (874.40) | (1,271.21-1490.19) | 0.275a |
| Annual number of prescriptions/patient | 127.9 (43.6) | (122.4-133.4) | 134.3 (50.4) | (128.0-140.6) | 0.228a |
| Prescriptions in generic drugs (%) | 39.1 (15.8) | (37.1-41.1) | 40.6 (16.4) | (38.5-42.65) | 0.299b |
| Prescriptions in new drugs (%) | 0.9 (2.7) | (0.6-1.2) | 0.7 (2.3) | (0.4-1.0) | 0.972a |
| 12 months after intervention | |||||
| Annual drug expenditure/patient (€) | 1,062.30 (802.70) | (961.79-1,162.81) | 1,211.30 (824.40) | (1,108.07-1,314.53) | 0.010a |
| Annual number of prescriptions/patient | 109.1 (40.6) | (104.0-114.2) | 118.5 (43.1) | (113.1-123.9) | 0.013b |
| Prescriptions in generic drugs (%) | 46.3 (17.1) | (44.2-48.4) | 45.5 (17.2) | (43.3-47.6) | 0.585a |
| Prescriptions in new drugs (%) | 1.1 (3.2) | (0.7-1.5) | 1.4 (3.0) | (1.0-1.8) | 0.064a |
| Before-after differences | |||||
| Reduction in drug expenditure (€) | 233.75 (510.46) | (169.83-297.67) | 169.40 (527.35) | (103.37-235.43) | 0.171a |
| Reduction in drug expenditure (%) | 14.3 (40.8) | (19.4-9.2) | 7.7 (42.7) | (13.0-2.35) | 0.041a |
| Reduction in number of prescriptions (%) | 12.5 (22.0) | (15,2-9,75) | 8.9 (23.4) | (11.8-2.3) | 0.091b |
| Increase in generic drug prescriptions (%) | 7.4 (11.7) | (5.9-8.8) | 5.1 (10.6) | (3.8-6.4) | 0.025b |
| Increase in new drug prescriptions (%) | 0.2 (3.0) | (−0.2-0.5) | 0.7 (2.4) | (0.4-1.0) | 0.007a |
95%CI: 95% confidence interval; SD: standard deviation.
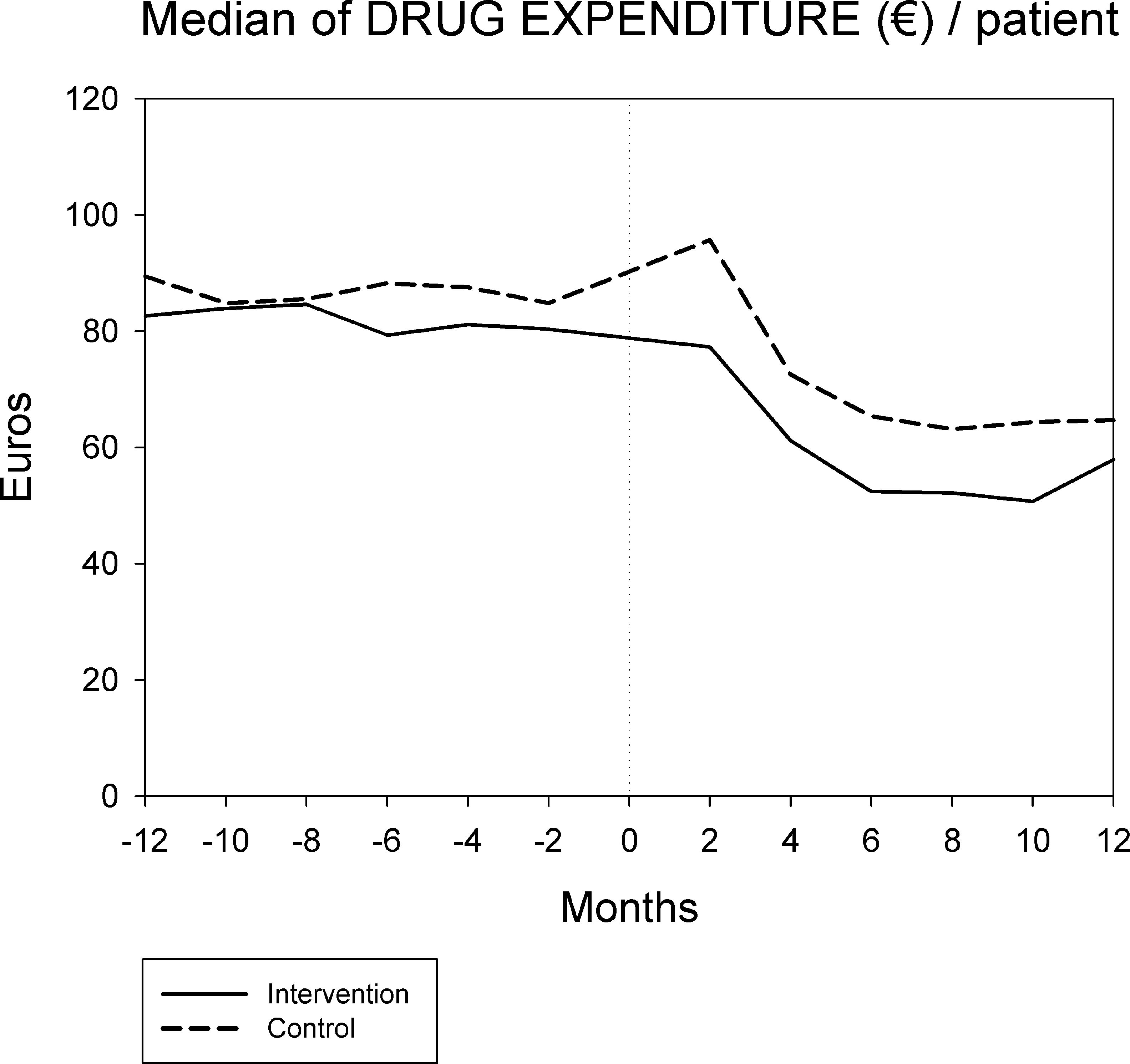
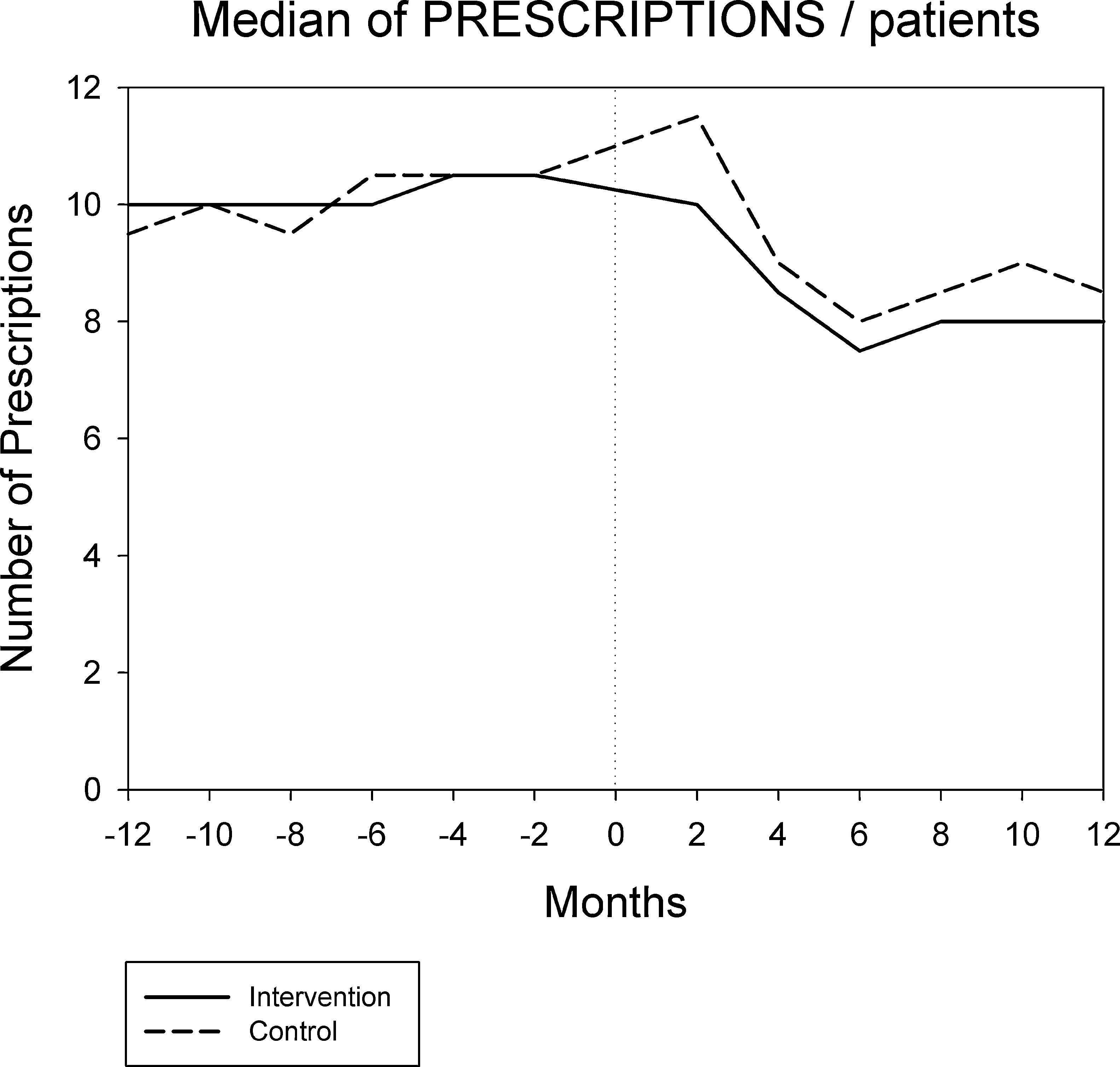
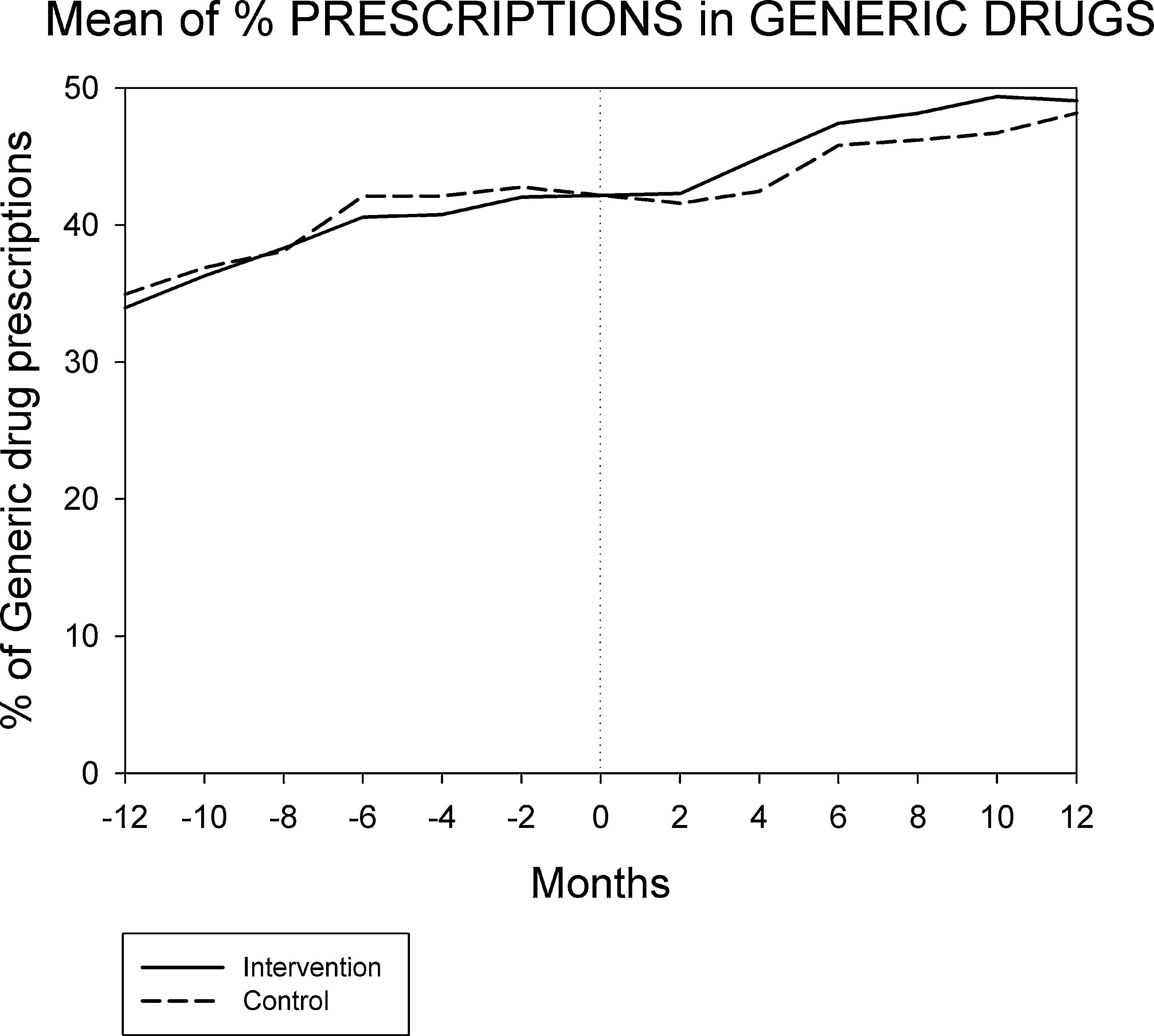
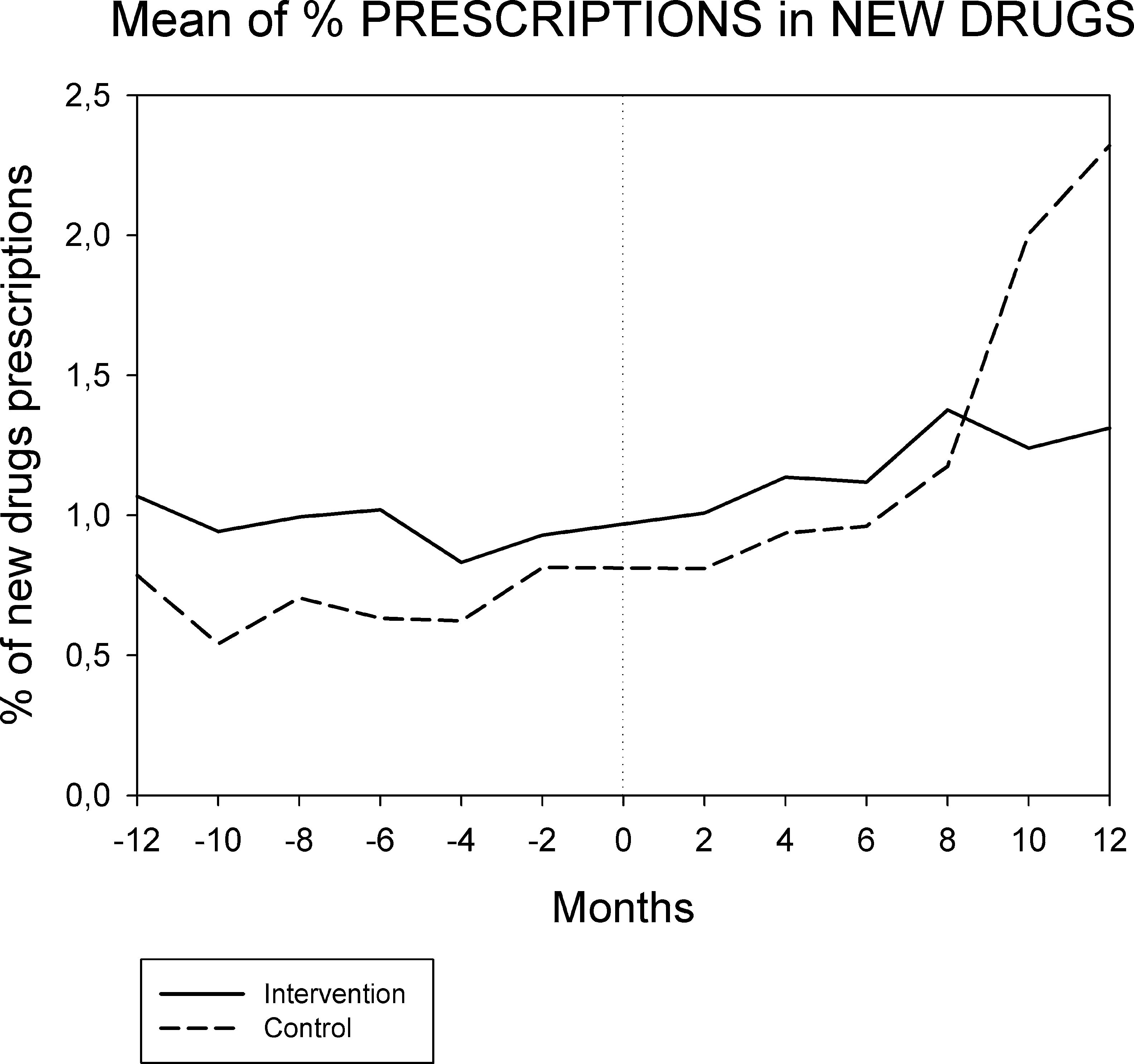
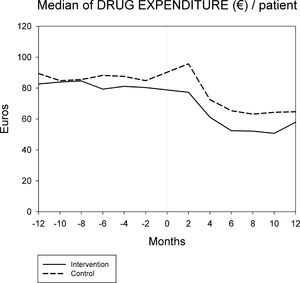
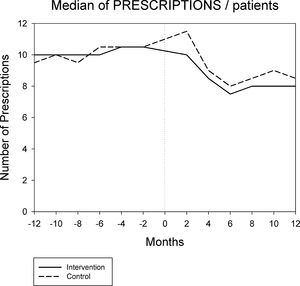
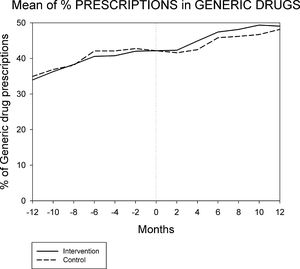
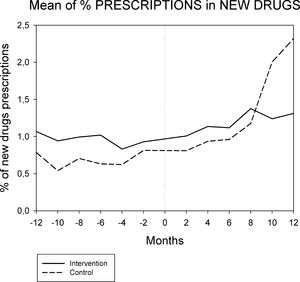
Figures 1 and 2 displays the monthly evolution of median drug expenditure and median number of prescriptions during 12 months pre-intervention and 12 months post-intervention for both groups. Further, the higher percentage of generic drugs prescribed in the intervention group remained largely unchanged throughout the one-year follow-up (Fig. 3). Finally, as regard the prescription of new drugs (Fig. 4), it is the standard care group that shows a rather more ascendant pattern, particularly over the last 4 months in the study.
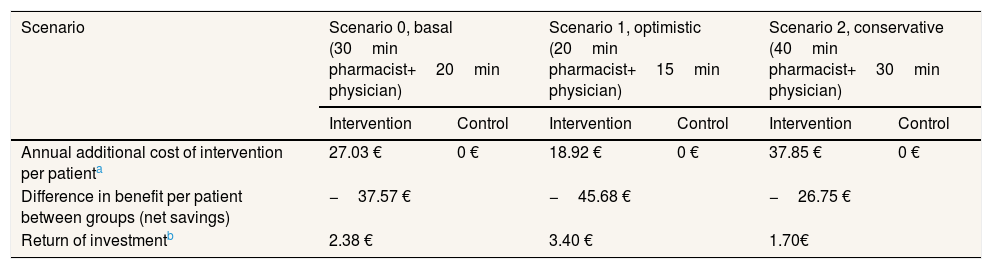
Table 2 presents the additional costs and savings under three scenarios. The average annual saving resulting from the study intervention is 37.57 € per patient a year. According to the sensitivity analysis performed, annual savings per patient range from 45.68 € (optimistic scenario) to 26.75 € per patient (conservative scenario). If we were to estimate the return on investment of the study intervention, dividing cost savings (64.30 €) by annual cost of intervention for each scenario, we would argue that for 1 € invested in the program we would be saving an average of 2.38 € per patient a year (ranging from 1.70 € to 3.40 €).
Sensitivity analysis of the annual benefits per patient considering three cost scenarios.
| Scenario | Scenario 0, basal (30min pharmacist+20min physician) | Scenario 1, optimistic (20min pharmacist+15min physician) | Scenario 2, conservative (40min pharmacist+30min physician) | |||
|---|---|---|---|---|---|---|
| Intervention | Control | Intervention | Control | Intervention | Control | |
| Annual additional cost of intervention per patienta | 27.03 € | 0 € | 18.92 € | 0 € | 37.85 € | 0 € |
| Difference in benefit per patient between groups (net savings) | −37.57 € | −45.68 € | −26.75 € | |||
| Return of investmentb | 2.38 € | 3.40 € | 1.70€ | |||
Our study reveals that a one-time pharmacist intervention in primary care allows for a 12 months accumulated reduction in total drug prescription and expenditure in community-dwelling polymedicated elderly people. It also shows an increase in the number of prescribed generic drug and a reduction in the number of prescribed “new drugs”. Since savings in drug expenditure are greater than costs derived from the intervention, the pharmacist intervention can be a cost-effective alternative to standard of care rendering a positive return on investment (2.38 €) per euro.
Our results are consistent with other findings from previous studies evaluating the economic impact of pharmacist interventions. In a series of systematic reviews conducted for the American College of Clinical Pharmacy, the economic value of clinical pharmacist services was summarized and evaluated. The first review, conducted by Schumock et al.,19 evaluated articles published from 1996-2000. They concluded that for every $1 invested in clinical pharmacy services, $4 was achieved in reduced costs or other economic benefits. A review by Pérez et al.,20 that included articles published between 2001-2005, showed similar results. The most recent published review on the topic21 incorporated fewer number of studies, published between 2006-2010, and could not provide a benefit/cost ratio as previous reviews did. One important difference between our study and other published studies included in these reviews is that the latter focused more on specific clinical conditions. Brennan et al. demonstrated a return on investment ratio of 3:1 using an integrated pharmacy program to improve medication prescription and adherence rates in diabetes patients.22 Pharmaceutical interventions have also proven to be cost-effective in pathologies such HIV,23 depression24 or hypertension.25 Despite these pieces of evidence, there are very few studies that focused on the economic evaluation of a pharmacist intervention that aim at reducing potentially inappropriate prescriptions and at improving drug appropriateness in community-dwelling polymedicated elderly people. When published, such studies showed very modest savings regarding medication costs with no statistical significance.26
Monthly evolution of median cost, median prescriptions per patient and percentage of generic drugs showed a somewhat parallel evolution on control and intervention groups indicating that the initial gap between both groups generated by the pharmacist intervention had a long-lasting effect on both variables. This effect appears to be slightly diluted one year after the intervention, and perhaps indicates that another pharmaceutical intervention should be done as reinforcement. On the other hand, “new drugs” showed an increase in prescription over the last 4 months in the study, pointing to a greater penetration of new products as therapeutic options in this group.
Although a significant reduction in drug prescription and expenditures was observed resulting from the intervention, it is of relevance to report that the control group also experienced a significant reduction in drug expenditure itself during the follow-up study period. This fact can be partially attributed to the study design, which was an open-label randomized clinical trial with possible intervention-to-control contagion. The prescribing physicians who received recommendations from the pharmacist regarding patients in the intervention group also visited patients in the control group. Consequently, the control group have indirectly benefited from the intervention, possibly diluting the true effect of the intervention. Moreover, the decrease in drug prescription in the control group may also be explained by other measures put forward by the Catalan Health Service to control drug expenditure in the primary care setting and by other important state control measures such as the Royal Decree-Law 16/2012, which introduced the co-payment system for outpatient pharmaceutical services and the exclusion of more than 400 products from public funding.
The reduction in total drug expenditure is largely a consequence of a decrease in the number of drugs prescribed (drug discontinuations), which accounts for 9.2% of total initial prescriptions. In addition, dose adjustments accounted for 6.9% and drug substitutions for 3.1% of initial prescriptions17. The use of generic drugs had a limited impact in drug cost savings since the Spanish drug regulation does not allow for differences in publicly financed prices between generic and non-generic drugs. Finally, it should be noted that although the prescription of new drugs is only a small percentage of overall drug prescription, the control group shows a more than three-fold increase in this respect when compared to the intervention group. Given the very high price of these new drugs, small differences in prescription frequency may have a relevant economic impact.
Probably, the main strength of the study is its controlled and randomized experimental design. There are a number of advantages of performing economic studies as part of an on-going clinical trial.27 Among other benefits, it is argued that since economic evaluation largely depends upon the quality of the data generated, clinical trials are an efficient setting for economic analyses. Both the quality of the data and the greater control over potential sources of bias favor clinical trials. However, the literature also outlines some limitations, the most relevant to our study being the generalization of the economic impact of the intervention in real-world practice. To overcome this limitation, we have selected a comparator that represents the most realistic choice in real practice. Other study limitations include: a) the intervention-to-control contagion, which may dilute the effect of the intervention; b) the exclusion of deaths from the analysis, which may carry an underestimation of total drug expenditure but do not alter inter-group comparisons because of the balanced number of deaths between groups; and c) cost elements considered are only cost in drug consumption and direct costs in human resources (time invested was a theoretical estimation), therefore more economic impact studies and full economic evaluations are required.
In summary, the present study shows that the intervention of a clinical pharmacist in the primary care setting evaluating all medication in polymedicated community-dwelling elderly subjects is responsible for a reduction of approximately 7% in drug expenditure. Such a community intervention may be a cost saving alternative with a possible positive return on investment.
Potentially inappropriate prescribing has been found to be frequent in elderly population and associated with morbidity, adverse drug events, hospitalizations, and health care expenditures. Several criteria and algorithms have been developed to reduce it. However, limited evidence exists about economic evaluations of these interventions.
What does this study add to the literature?The intervention of a clinical pharmacist in the primary care setting evaluating all medication in polymedicated elderly subjects is responsible for a reduction of approximately 7% in total drug expenditure (a mean reduction of 64 € per patient a year). Such a community intervention is a cost saving alternative with a positive return on investment.
Miguel Ángel Negrín Hernández.
Transparency declarationThe corresponding author on behalf of the other authors guarantee the accuracy, transparency and honesty of the data and information contained in the study, that no relevant information has been omitted and that all discrepancies between authors have been adequately resolved and described.
Authorship contributionsL. Campins designed the study, contributed in the fieldwork and wrote the manuscript. M. Serra-Prat designed the study, performed the statistical analysis and wrote the manuscript. E. Palomera performed the statistical analysis and review the manuscript. I. Bolibar review the manuscript with important contribution in methodological aspects. M.A. Martínez, medical coordinator of the study, contributed in the fieldwork and review the manuscript. P. Gallo review the manuscript with important contribution in economic aspects.
The authors thank the study participants and ICS (Institut Català de la Salut) and CSdM primary healthcare professionals who assisted with the study. REMEI Group members who participated in the fieldwork: Clara Agustí, Miren Maite Aizpurua, Maria Alegre, Antònia Armada, Azhara Sánchez, Mireia Bancells, Eugènia Barbena, Maria Bartolomé, Montserrat Bosch, Isaac Buxadé, Mateu Cabré, Marta Calvo, Lluís Campins, Marcel·la Camps, Dolors Casabella, Toni Casanova, Gemma Casas, Rosa Castellanos, Salvador Castro, Berta Chaves, Sílvia Cid, Rosa Mª Coma, Enric Corona, Joan Domenech, Imma Esteva, Esther Fabré, Xavier Fabregas, Pere Flores, Isabel Font, Consol Garcia, Vanesa Garcia, Teté González, Inés Gozalo, Teresa Gros, Teresa Gurrera, Gregorio Hinojosa, Núria Jerez, Mercè Jiménez, Tamara Jiménez, Josep Juanola, Lucia Jurado, Esther Limón, Pere Lledonet, Jordi Lloret, Montse Lloret, Ainhoa López, David López, Carolina Luna, Mozgham Mahramci, Vanessa Marta, Juan José Martí, Oriol Martí, Dolors Martínez, Miquel Àngel Martínez, Mireia Massot, Laura Mateu, Núria Mengual, Nacho Menjón, Mar Mir, Juan Carlos Montero, Pilar Montero, Susana Morales, Josep Nuñez, Ana Palacio, Elisabet Palomera, Mercè Palomera, Mònica Papiol, Joan Pascual, Lourdes Pató, Miquel Robusté, Ingrid Roca, Ana Roces, Mel Roger, Jordi Salabarnada, Fernando Samaniego, Dolors Sánchez, Elisa Sanz, Miriam Serra, Pere Serra, Mateu Serra-Prat, Cristina Serrano, Dorte Skiffter, Clara Soler, Pelin Son, Josep Sorribes, Pere Toran, Eulàlia Torrellas, Carles Tria, Yolanda Verde and Isabel Villarroya.
FundingThis project was funded by a grant from the Spanish Ministry of Health (Independent Health Research Ref. EC11-313) and a grant from the Catalan Government Health Service (SLT/682/2012). These governmental funding bodies played no role in study design, data collection, analysis and interpretation or in the decision to approve publication of the finished manuscript.
Conflicts of interestsNone.